Home mechanical ventilation in children with chronic respiratory failure: a narrative review
Article information
Abstract
Advances in perinatal and pediatric intensive care and recent advances in mechanical ventilation during the last two decades have resulted in an exponential increase in the number of children undergoing home mechanical ventilation (HMV) treatment. Although its efficacy in chronic respiratory failure is well established, HMV in children is more complex than that in adults, and there are more considerations. This review outlines clinical considerations for HMV in children. The goal of HMV in children is not only to correct alveolar hypoventilation but also to maximize development as much as possible. The modes of ventilation and ventilator settings, including ventilation masks, tubing, circuits, humidification, and ventilator parameters, should be tailored to the patient’s individual characteristics. To ensure effective HMV, education for the parent and caregiver is important. HMV continues to change the scope of treatment for chronic respiratory failure in children in that it decreases respiratory morbidity and prolongs life spans. Further studies on this topic with larger scale and systemic approach are required to ensure the better outcomes in this population.
Introduction
Advances in perinatal and pediatric intensive care have led to an increasing number of patients with complex medical conditions surviving. For instance, the number of children with chronic respiratory failure requiring long-term mechanical ventilation has increased exponentially worldwide [1-3]. In addition, recent advances in home mechanical ventilation (HMV) during the last two decades have enabled children who would previously have been confined to living in the hospital to return home and participate in family and community activities [4-7]. Moreover, the diseases requiring long-term mechanical ventilation have expanded from respiratory diseases such as chronic lung disease to neuromuscular disease, craniofacial abnormalities, spinal cord injuries, and central hypoventilation syndromes [8,9].
There are two major types of ventilators, namely regular intensive care unit (ICU) ventilators and HMVs. While regular ICU ventilators are more powerful and have more parameters that can be controlled according to the patient’s needs (e.g., trigger type, trigger sensitivity, slope of pressurization, and cycling criteria) as well as monitoring functions, they are large and expensive and require more knowledge and skills to operate. In contrast, HMVs are portable devices that are easier to use and require fewer resources but have more technical limitations because they cannot support a higher level of pressure (>20 cmH2O) and cannot maintain a higher oxygen fraction (FiO2 of >60%). Therefore, HMVs are more suitable for patients with chronic respiratory failure than for patients with hypoxemic respiratory failure [10]. Although HMV is now accepted as a standard treatment option for patients with chronic respiratory failure, patient selection and HMV management vary substantially among countries [11]. Moreover, for children, HMV in children has more considerations than for adults because children are still developing physically and mentally. This review outlines patient selection and special considerations for HMV in children, its modes and technical setup, follow-ups, monitoring and weaning, and potential risks of HMV before finally describing caregiver burden and education.
Diagnosis and patient selection for home mechanical ventilation
Cellular metabolism in humans requires continuous oxygen supply and carbon dioxide removal. The respiratory system comprises two independent subsystems: the gas exchange system (the lungs) and the ventilatory system (the respiratory pump) [12]. Respiratory failure is defined as a problem in either (or both) of these systems and often arises from an imbalance between the respiratory workload and ventilatory strength [13]. Its diagnosis is based on the arterial blood gases (ABGs), arterial partial pressure of oxygen (PaO2) less than 60 mmHg, and alveolar partial pressure of carbon dioxide (PaCO2) greater than 50 mmHg, which correspond to the hypoxemic (or type I) respiratory failure and hypercapnic (or type II) respiratory failure, respectively [14]. Hypoxic respiratory failure is treatable with oxygen therapy, whereas hypercapnic respiratory failure requires ventilatory support. Cases of multiple disorders or many acute respiratory failures require a combination of ventilatory support and oxygen supplementation.
The respiratory pump is a complex system. Breathing is regulated by the respiratory center of the medulla oblongata. Respiratory signals are transmitted via the central and peripheral neural tracts to the neuromuscular endplate of the respiratory muscles, including the diaphragm and intercostal muscles, sternocleidomastoid, and pectoral muscles, and generate inhalation by the negative pressure in the chest wall. In contrast, exhalation is normally passive, caused by the elastic recoil of the chest wall and the lung parenchyma. As respiratory failure results from the imbalance between the respiratory workload and ventilatory strength (Fig. 1) [15], diseases for which HMV is necessary in children can be classified into three categories, according to the physiological abnormalities they cause (Table 1) [9,16].

Respiratory balance. (A) Normal respiratory balance, in which the load imposed on the respiratory system, the capacity of the respiratory muscles, and the central drive are in equilibrium. (B) A decrease in central drive (dotted line) causes a decrease in respiratory muscle activity and, subsequently, a reduced alveolar ventilation. (C) A weakness of the respiratory muscles or an increase in respiratory load causes an increased central drive (bold arrow). Alveolar ventilation occurs when the imbalance exceeds a specific threshold. (D) Noninvasive ventilation can correct disequilibrium in the respiratory balance by replacing the central drive, unloading (in case of an increase in respiratory load, as shown), or assisting the respiratory muscles (in case of respiratory muscle weakness). Reprinted from Amaddeo et al. [15] with permission from Elsevier.
Collecting the medical history and performing clinical examinations are the first steps in the diagnosis of chronic respiratory failure. The signs and symptoms suggestive of chronic respiratory failure include sleep disturbances such as frequent awakening during sleep, unrestful sleep, daytime somnolence, and nightmares; worsening of accompanying symptoms of the underlying disease such as weight loss, dyspnea, and decreased exercise capacity; polycythemia; signs of carbon dioxide-associated vasodilation, such as conjunctival injection, leg edema, and morning headache; tachypnea; tachycardia; and anxiety or personality change [16]. In addition, respiratory muscle strength should be measured as an instrument-based assessment; however, this is not always possible, especially in small children or older children with mental retardation. Consequently, the ventilation and gas exchange status must be directly assessed using the arterial PCO2 via blood gas analysis or continuous monitoring using transcutaneous PCO2 monitoring (PTcCO2), alternatively. PTcCO2 better captures the complete ventilation trend, despite the possible deviation of individual values from those obtained by the ABG analysis, which is the gold-standard diagnostic test [17].
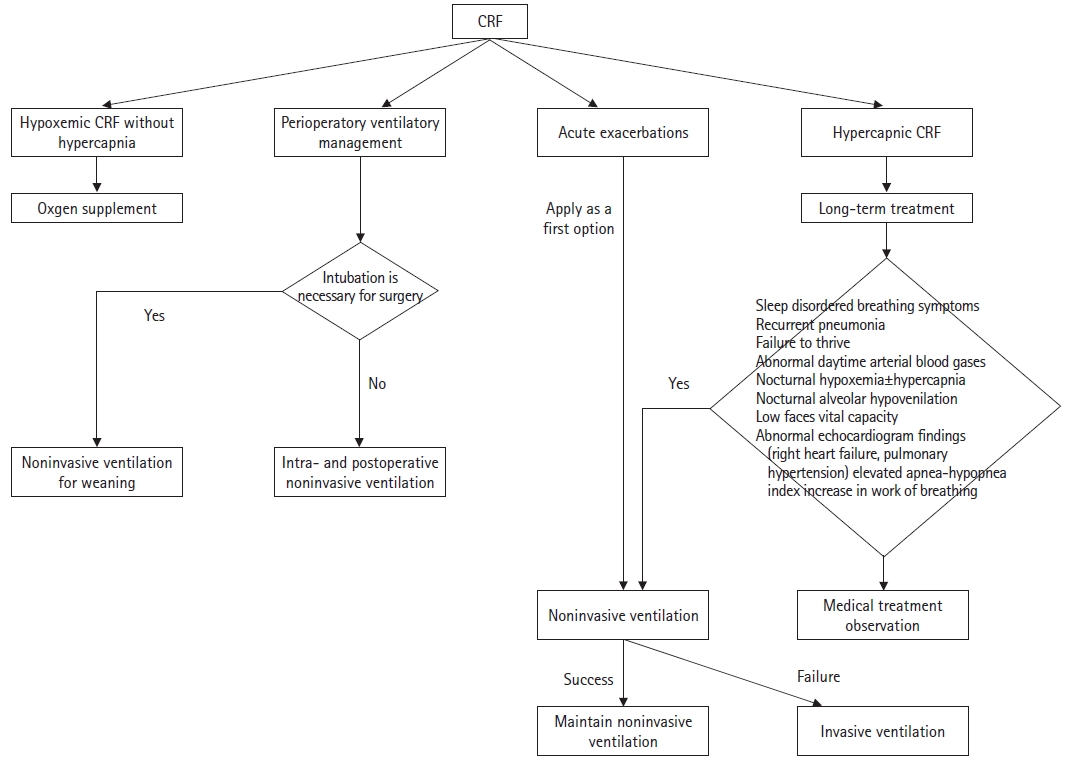
Chronic respiratory failure can only be treated with ventilator support using a mechanical ventilator, either noninvasively or invasively. Fig. 2 shows the application algorithm for noninvasive and invasive ventilation in children [11]. The goal of HMV in children with chronic respiratory failure is to correct alveolar hypoventilation and maximize development, as possible [18]. The effects of long-term mechanical ventilation, including improving alveolar ventilation, alleviating subjective symptoms of chronic respiratory failure, improving blood gases, reducing morbidity and mortality, and enhancing the quality of life of the affected children, are well established in the previous studies; however, these studies were largely observational and limited to certain disease entities (e.g., congenital central hypoventilation syndrome or progressive neuromuscular diseases such as Duchenne muscular dystrophy) [19-29]. However, in other diseases for which HMV might be required, similar effects can be expected, provided that there is no parenchymal lung disease component in the chronic respiratory failure [16].
Special considerations for home mechanical ventilation in children
HMV in children is much more complex than that in adults. First, smaller children and older children with mental retardation might not be able to communicate or cooperate with assessment and/or treatment. In addition, there is a risk of accidental removal of the tracheostomy tubes. Second, children requiring HMV often have various comorbidities; therefore, they should ideally be cared for by a multidisciplinary team for the underlying conditions. Third, the airway diameters are smaller than those in adults, which makes children more vulnerable to airway obstruction due to secretion when airway clearance is compromised. Fourth, when using noninvasive ventilation (NIV), children unable to remove the ventilation mask themselves might be endangered in cases of vomiting or equipment errors. Finally, care should be taken to ensure that the child can develop normally to the possible extent in terms of speech/language development as well as facial bone growth. Poorer developmental outcomes have been reported in children on HMV compared to those in healthy controls [30,31], which may arise from a combination of factors including skeletal muscle weakness due to underlying diseases or prolonged bed rest in the acute phases of illness, reduced functional status due to ventilator dependency, increased metabolism due to chronic respiratory distress, and prolonged supine positioning during infancy [30]. Although rehabilitation strategies have been proposed [31,32], no clinical study has examined the effectiveness of rehabilitation therapy in this population. Further studies that focus on the developmental aspect of children on HMV are warranted to facilitate the normal development of these children as far as possible. In infants and small children who have undergone tracheostomy, cannulae with adequate leakage via the glottis should be selected for vocalization, and speaking aids should be implemented where possible. A recent study conducted in a developing country setting reported that having parents as the primary caregiver was related to better speech and language skills in these children [30], which may underscore the importance of the role of primary caregivers in development. As facial bone growth occurs in the early years of life, NIV may hinder the normal facial bone growth, resulting in mid-facial deformity (flattening of mid-face and maxilla retrusion) due to the high pressure applied on the facial bone for a relatively large part of the day in young children, even though NIV is only used during sleep; to reduce this risk, alternative use of different masks that apply pressure to different parts of the face can be considered [33]. In addition, a higher contact pressure is associated with an increased risk of facial deformity, thus, masks of young children should be monitored for optimal fit.
In addition, several technical issues are associated with HMV in children. Because small children might not generate sufficient inspiratory flow to trigger a ventilator, HMV for children should have a sensitive trigger. In addition, ventilation volumes can be very low in infants and small children, and the breathing patterns of the children can be irregular in terms of the frequency and depth of breathing. Therefore, home mechanical ventilators for children should be able to generate a low tidal volume, and HMV with pressure presets may be more appropriate than HMV with volume presets because they can better adapt to irregular breathing patterns [16].
Modes of home mechanical ventilation in children
HMV can be delivered either noninvasively or invasively. NIV uses nasal masks, oronasal masks, full-face masks, or mouthpieces, whereas invasive ventilation requires tracheostomies. The choice of these modes does not depend on a particular disease but rather on the type and severity of the respiratory failure. In general, obstructive sleep apnea syndrome, central respiratory regulation disorder, chronic alveolar hypoventilation, and other chronic respiratory failures are all candidates for NIV. Tracheostomies may result in significant complications, such as granuloma, infection, obstruction of the cannulae, and accidental decannulations, which are more common in children than in adults [34]. Moreover, tracheostomy may lead to impaired language development and negatively impact the body image of children. Therefore, NIV should always be considered before tracheostomy, as is also the case for adults. However, several conditions require tracheostomy insertion, including congenital malformation syndromes with obstruction of the upper airways, vocal cord paralysis, subglottic stenosis, tracheomalacia, progressive neurological diseases with bulbar palsy, bronchopulmonary dysplasia, pulmonary hypoplasia, and a ventilation duration of >16 hours per day [35]. In some of these conditions, such as some of the congenital malformation syndrome, subglottic stenosis, tracheomalacia, and bronchopulmonary dysplasia, tracheostomy may be closed later in childhood [36]. The advantages and disadvantages of NIV are summarized in Table 2 [37,38]. In cases of NIV failure, tracheostomy is the only alternative and the ultimate therapeutic option [11].
1. Noninvasive ventilation
1) Unintentional air leakage
While a certain degree of air leakage is inevitable in NIV because it is not a completely closed system, and it does not necessarily compromise the effectiveness of NIV if the amount of air leakage is small [37]. However, a large amount of air leakage compromises the ventilation effectiveness and may result in nasal mucosal drying and asynchrony between the patient and the ventilator as the ventilator perceives it as an attempt to inhale and continues to dispense breath even though the patient has completed inhalation and is ready to exhale [37]. Therefore, monitoring the delivered tidal volume and air leakage is important in patients undergoing NIV, and most home ventilators have leakage compensation mechanisms [39].
2) Interfaces for children
Various types of ventilation masks are used for NIV, but none can be perfectly sealed, at least intermittently. Therefore, the ventilators must generate a high flow to compensate for leakage to reach preset pressure. Arguably, the most important factor determining successful NIV application is the choice of an appropriate mask that minimizes leakage and maximizes patient’s comfort [40]. The masks currently being used in children include follows: nasal pillows or nasal plugs that occlude the nostrils; nasal masks, which cover the nose; oronasal masks, which cover the mouth and the nose; full-face masks, which cover the mouth, the nose, and the eyes; mouthpieces or oral masks; and the helmet, which cover the entire head. Nasal masks provide greater patient comfort and cause fewer skin injuries, making them the most commonly used masks [41]. Nasal masks are preferred in small children because there is less static dead space, they cause less claustrophobia, and they allow communication and expectoration. Oronasal masks and full-face masks must be reserved for use only when nasal masks are not effective due to oral leakage during sleep as they have potential risks of aspiration in cases of gastroesophageal reflux or nausea/vomiting. Mouthpieces can be used for children with neuromuscular disease requiring extended ventilation support in daytime, in addition to nocturnal ventilation [42]. Helmets are effective interfaces for small children, and they are commonly used in the pediatric ICU (PICU). However, their large dead space and the risks of asphyxia in case of technical failure of the ventilator make them unsuitable for HMV [43-45]. The following should be considered when selecting an appropriate mask: (1) patient age and weight, (2) facial bone and the skull anatomy, (3) the presence of mouth breathing, (4) patient's ability to remove the mask by himself/herself, (5) patient comfort and the level of unintentional leakage, and (6) the patient tolerance regarding skin injury or facial deformity [46].
2. Invasive ventilation
1) Tracheostomy and tracheal cannulae
Tracheostomy should be stable when starting HMV; therefore, elective surgical tracheostomy for HMV is always preferred to percutaneous dilatational tracheostomy. Once the tracheostomy stoma is healed, it can be exchanged by a trained nurse alone and even by trained nonprofessional caregivers [47]. In cases of sudden cannulae blockage or a difficult cannulae change, spare cannulae of the same size and smaller should always be prepared. Before discharge from the hospital, parents or caregivers should learn how to manage a tracheostomy, identify complications, and perform cardiopulmonary resuscitation in case of an emergency [47].
2) Humidification and warming
During invasive ventilation, humidification and warming of the inhaled air are necessary to prevent drying of the bronchial mucosa and thickening of secretions [48]. In addition, children tend to have relatively high respiratory rates and, therefore, a higher fluid loss via the airway, which increases the importance of sufficient level of humidification in this population.
Humidification and warming can be achieved either through heat and moisture exchanger (HME) filters or via active humidification. The use of HME filters has the advantage of simplicity; however, they may increase the work of breathing and negatively impact CO2 elimination. In particular, when using speaking aids, uncuffed cannulae, or fenestrated tracheal cannulae, HME filters are ineffective because air escapes to the larynx, thus requiring active humidification, although this is less convenient [49,50]. Active humidification during the night and HME filters during the day are generally considered to be acceptable [16].
Technical setup of home mechanical ventilator
1. Tubings and circuits
There are two types of tube systems: single-tube systems (single-limb circuits) and double-lumen tube systems (double-limb circuits). While double-lumen tube systems (one tube for inspiration and the other for expiration) are mostly used in acute care settings, such as ICUs, single-tube systems are most commonly used in HMV because they are less cumbersome. When using single-tube systems, unintentional leaks and tidal volumes are estimated instead of measured; therefore, if it is necessary to measure expiratory volume accurately, a double-lumen tube system should be used, which is rarely the case in this population [51-53].
2. Pressure versus volume preset
Pressure-preset mechanical ventilation is more widely used than volume-preset ventilation. Pressure-preset mechanical ventilation delivers airflow according to a predefined positive pressure in the airways for a given time. In contrast, volume-preset ventilation provides a fixed volume during a given time regardless of the airway pressure. The advantage of volume-preset ventilation is the delivery of a constant tidal volume, assuring that the desired minute ventilation is achieved; however, the major disadvantage of this mode is the fixed ventilatory support that does not allow changes with the varying demands of the patient. Although the two modes do not differ in terms of NIV effectiveness, pressure-preset ventilation is associated with fewer side effects, such as flatulence and gastric distension [54-56]. In addition, as stated earlier, pressure-preset ventilation is preferred in children because it can better adapt to their irregular breathing patterns.
If a patient experiences problems under a particular ventilation mode, another ventilation mode should be attempted during hospitalization and close monitoring.
3. Ventilator alarms
Alarms in NIV are based on clinical practice rather than scientific evidence due to the lack of studies examining their clinical utility, especially for HMV [57]. Mask interfaces in NIV cannot be completely sealed, which may cause frequent and often spurious alarms. Therefore, careful setting of the alarm system is necessary to ensure that it functions only when there is a genuine need [57,58]. An alarm system is mandatory in invasive ventilation because the blockage of cannulae may lead to a significant risk to the patient. Patients with a speaking valve, uncuffed cannulae, or fenestrated cannulae require an alarm setting that can detect the disconnection and/or hypoventilation, as recommended in adults [58].
Home mechanical ventilation follow-up and monitoring
1. Pulse oximetry
A pulse oximeter is necessary during the invasive ventilation; continuous pulse oximetry monitoring is mandatory in children with tracheostomy, as well as in children using full-face masks or oronasal masks, who cannot remove the masks by themselves. Because frequent false alarms might distract caregivers from concentrating on real alarms, the use of appropriate adhesive sensors and an oximeter with an artifact-minimizing function should be adopted to minimize false alarms.
Oxygen saturation may decrease in cases of airway infection, increased secretion, coughing, dyspnea, and enforced breathing. When the SpO2 is greater than 95%, no intervention is required; for SpO2 values are between 90% and 95%, readjusting interfaces and/or secretion clearance should be attempted, after which further treatment can be sought in an outpatient setting; and if SpO2 is less than 90% despite HMV, immediate action must be taken (e.g., visiting emergency department or calling for the ventilation center, etc.) [16].
Of note, oxygen alone should not be used to correct decreased SpO2 values in children with alveolar hypoventilation due to neuromuscular diseases because it can worsen hypercapnia, mask the progression of underlying problems, and delay diagnosis and appropriate treatment [59,60].
2. Follow-ups for home mechanical ventilation in children
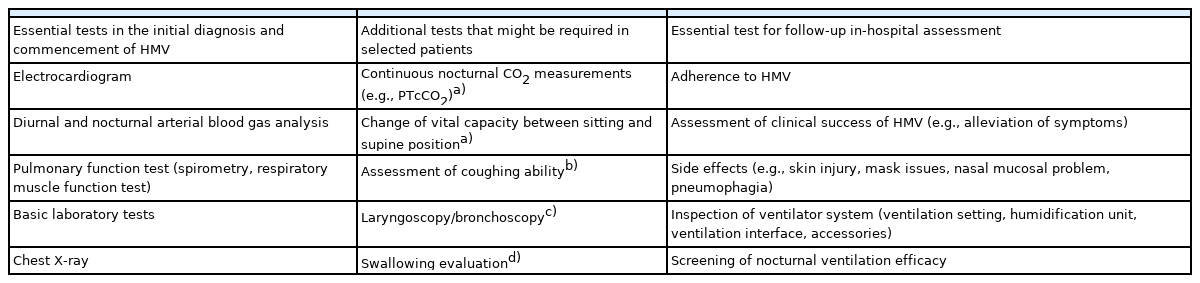
Regular follow-ups are required to ensure successful HMV in children as HMV treatment should be adapted to the growth of the child and the progression/improvement of the underlying disease. Despite marked heterogeneity in the provision of HMV services, costs, and healthcare system across countries as well as in the individual factors such as underlying disease, medical stability, the age of the child, and socioeconomic status, follow-up in-hospital assessment including nocturnal CO2 monitoring and synchronization of ventilation is generally recommended to be performed 1 to 3 months after commencing HMV for the first time [61]. Subsequently, repeating these assessments at 6-month intervals is recommended. Tracheoscopy should be performed at least once annually in invasively ventilated children to adjust the tube size and assess for complications such as tracheal granuloma or ulcers [16]. The follow-up assessment of HMV should include the tests summarized in Table 3.
Tests that should be performed during the initial diagnosis, commencement of HMV, and follow-up in-hospital assessment of children undergoing HMV
Any changes made to the ventilators and their setup, including ventilator machine (even from the same manufacturer), ventilation interface (masks and circuits), oxygen application system, humidification system, ventilation parameters, and tracheal cannulae models, can substantially influence ventilation efficacy and should only be performed under thorough monitoring of the attending physician in in-hospital settings [62-66].
Weaning
A recent international consensus statement by the European Respiratory Society indicated weaning from continuous positive airway pressure (CPAP) or NIV in 6% to 40% of children [10]. Successful weaning is attributable to the physiological growth, weight loss, or interventions such as adenotonsillectomy, upper airway surgery, maxillofacial surgery, or orthodontic treatment [67-70]. Successful weaning in children with NMD is less common than that in children with other conditions [71-73]; however, this is expected to be changed along with the development of new therapeutic options, particularly in children with spinal muscular atrophy [11]. Currently, there are no established weaning criteria; however, a recent study by Mastouri et al. [73] proposed respiratory criteria for weaning from CPAP/NIV, including four major criteria and three minor criteria; all four major criteria must be fulfilled with at least two minor criteria for considering weaning from CPAP/NIV. The major criteria were: (1) resolution of nocturnal and daytime symptoms of sleep-disordered breathing after several nights without CPAP/NIV, (2) <2% of the recording time spent with a SpO2 of ≤90%, (3) <2% of recording time spent with a PTcCO2 of ≥50 mmHg, and (4) obstructive apnea-hypopnea index of <10 events/hr on a polysomnography or polygraphy. The minor criteria were: (1) minimal SpO2 of >90%, (2) maximal PTcCO2 of <50 mmHg, and (3) 3% oxygen desaturation index of ≤1.4 events/hr [73]. Even after successful weaning, recurrence of chronic respiratory failure is possible, depending on the underlying condition; therefore, continuous follow-up is necessary [73].
Potential risks of home mechanical ventilation
Despite the advances in newer ventilators and monitoring devices, children on HMV are still exposed to potential risks, with mortality in this group as high as 43% and medical complications occurring in up to 70% of children [74-76]. In addition, a recent study conducted in the United States analyzing rapid-response (RR) events and code events in children on HMV via tracheostomies in a non-ICU respiratory unit found that children on invasive ventilation via tracheostomy had 8.73 RR events per 1,000 patient days compared to 4.61 RR events per 1,000 patient days in all other hospitalized children. Similarly, children on HMV had 3.14 code events per 1,000 patient days compared to 0.74 in all other hospitalized children [77]. The causes of mortality have not been evaluated systemically; however, a previous study reported that only 34% of mortalities resulted from the progression of underlying diseases and 49% of mortalities were unexpected [75]. The potential risks in children on HMV include ventilator failure, airway obstruction by secretion, tracheostomy-related complications (accidental decannulation, bleeding, infection, etc.), acute lower respiratory tract infection, feeding problems, seizures, dehydration, and electrolyte imbalance [78,79]. A retrospective study reported that children with invasive mechanical ventilation were 16 times more likely to visit hospitals before scheduled visits than children with NIV (odds ratio, 16.3; 95% confidence interval, 2.1–127.4, p=0.008) [78]. Other risk factors for unscheduled hospital admissions were a shorter duration of caregiver education before discharge, a change in the management of the child within 7 days before discharge, and younger age [78-80]. A recent study in Germany evaluating differences in the incidences of complications between children with invasive ventilation at home and children with invasive ventilation at specialized nursing facilities found that children on invasive ventilation at home were more likely to experience tracheostomy-related incidents and infection compared to children with tracheostomy and long-term ventilation support at specialized nursing care facilities, while there was no difference in the incidence of potentially life-threatening severe adverse events with rapid deterioration of vital signs requiring immediate medical intervention, and may require resuscitation and hospital admission [81]. To reduce preventable adverse events in this population, well-organized caregiver education, providing home nursing services, and monitoring patients using telemedicine were proposed, in addition to training staff caring for children on home mechanical ventilators in the respiratory care unit [74,76-78,80,82].
Caregiver burden and education
1. Caregiver burden
Although HMV allows a more normal lifestyle compared to formal institutionalized (or hospital-based) ventilation treatment, it places significant burdens on patients and their caregivers, ranging from physiological and psychological to social, and existential issues. A recent systematic review of qualitative studies on users’ experiences of HMV reported that the users (namely, the patients and their caregivers) perceived that HMV increased their quality of life and permitted a more community- and home-based lifestyle. However, they also experienced continued worries and uncertainty as well as undermined autonomy and self-determinism. The caregivers also expressed difficulties in the collaboration between home-care assistants, including nurses, social-care assistants, and healthcare assistants [83]. Children on HMV and their caregivers have a significantly lower quality of life than healthy controls [30,31,84]. Nevertheless, a recent questionnaire survey revealed that although children with neuromuscular diseases on long-term ventilation treatment experienced a lower health-related quality of life and mental health, ventilator use did not show an additional negative impact on the quality of life [85]. Thus, care should be taken to enable a patient-centered treatment and care in this population and to reduce caregiver burden.
2. Patient and caregiver education
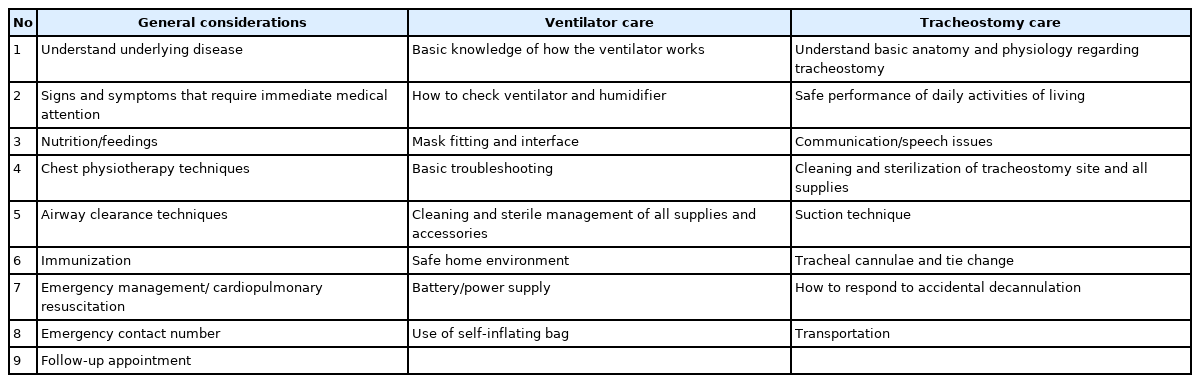
To ensure effective HMV in children, education for the patients (if possible) and caregivers is of critical importance. Although HMV enables a more normal daily life for the children compared to the formal institutionalized or hospital-based ventilation treatment, HMV presents many psychological, physiological, and social challenges to children and their families. In particular, children on HMV are dependent on support for up to 24 hours a day; thus, training for the caregivers and family members is very important. The essential topics for caregiver education are summarized in Table 4 [11,58,86]. Caregiver education totaling <14 days was associated with higher chances of nonscheduled hospital admissions in children on HMV [78]. The American Thoracic Society clinical practice guidelines recommend training periods of “several weeks before discharge” for children with invasive ventilation [86], while the Canadian Thoracic Society clinical practice guidelines recommend ≥2 weeks for patients with invasive ventilation and 48 hours for patients with NIV [87].
Conclusion
HMV continues to change the scope of treatment for chronic respiratory failure in children in that it decreases respiratory morbidity and prolongs life span. However, there remains a gap in the clinical practice and scientific evidence regarding the validated criteria for the initiation of HMV, optimal follow-up, and monitoring, and weaning criteria in different underlying diseases. Therefore, future studies are much needed. In addition, considering that the ultimate goal of HMV in children is not only to correct alveolar hypoventilation but also to maximize development as much as possible, future research should focus on the strategies to promote normal child development in every aspect, as well as provide optimal patient-centered care for the children and their caregivers.
Clinicians should select the best option for HMV according to the patient’s age, underlying disease, and HMV tolerance. Training for parents and caregivers is an essential part of HMV, and regular follow-up and good multidisciplinary care should be ensured.
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Funding
None.