Stent graft treatment of an ilioenteric fistula secondary to radiotherapy: a case report
Article information
Abstract
Fistulas between the arteries and the gastrointestinal tract are rare but can be fatal. We present a case of an ilioenteric fistula between the left external iliac artery and sigmoid colon caused by radiotherapy for cervical cancer, which was treated with endovascular management using a stent graft. A 38-year-old woman underwent concurrent chemoradiotherapy for cervical cancer recurrence. Approximately 9 months later, the patient suddenly developed hematochezia. On her first visit to the emergency room of our hospital, computed tomography (CT) images did not reveal extravasation of contrast media. However, 8 hours later, she revisited the emergency room because of massive hematochezia with a blood pressure of 40/20 mmHg and a heart rate of 150 beats per minute. At that time, CT images showed the presence of contrast media in almost the entire colon. The patient was referred to the angiography room at our hospital for emergency angiography. Inferior mesenteric arteriography did not reveal any source of bleeding. Pelvic arteriography showed contrast media extravasation from the left external iliac artery to the sigmoid colon; this was diagnosed as an ilioenteric fistula and treated with a stent graft. When the bleeding focus is not detected on visceral angiography despite massive arterial bleeding, pelvic arteriography is recommended, especially in patients with a history of pelvic surgery or radiotherapy.
Introduction
Arterioenteric fistulas are uncommon but life-threatening emergencies that require immediate intervention. They connect large arteries and the gastrointestinal tract and can be categorized into primary and secondary forms. Primary arterioenteric fistulas are extremely rare with an incidence of approximately 0.07% in the general population, as revealed by a large autopsy series [1]. Surgery is the standard treatment for arterioenteric fistulas. However, in emergencies, endovascular treatments such as stent graft exclusion can be applied. In the current report, we present the case of a patient with a primary arterioenteric fistula between the left external iliac artery and sigmoid colon caused by radiotherapy, which was managed successfully with endovascular treatment using a stent graft.
Case
Ethical statements: This study was approved by the Institutional Review Board (IRB) of Pusan National University Yangsan Hospital (IRB No: 05-2021-071) in accordance with the Declaration of Helsinki, and the requirement for informed consent from the patient was waived by the IRB.
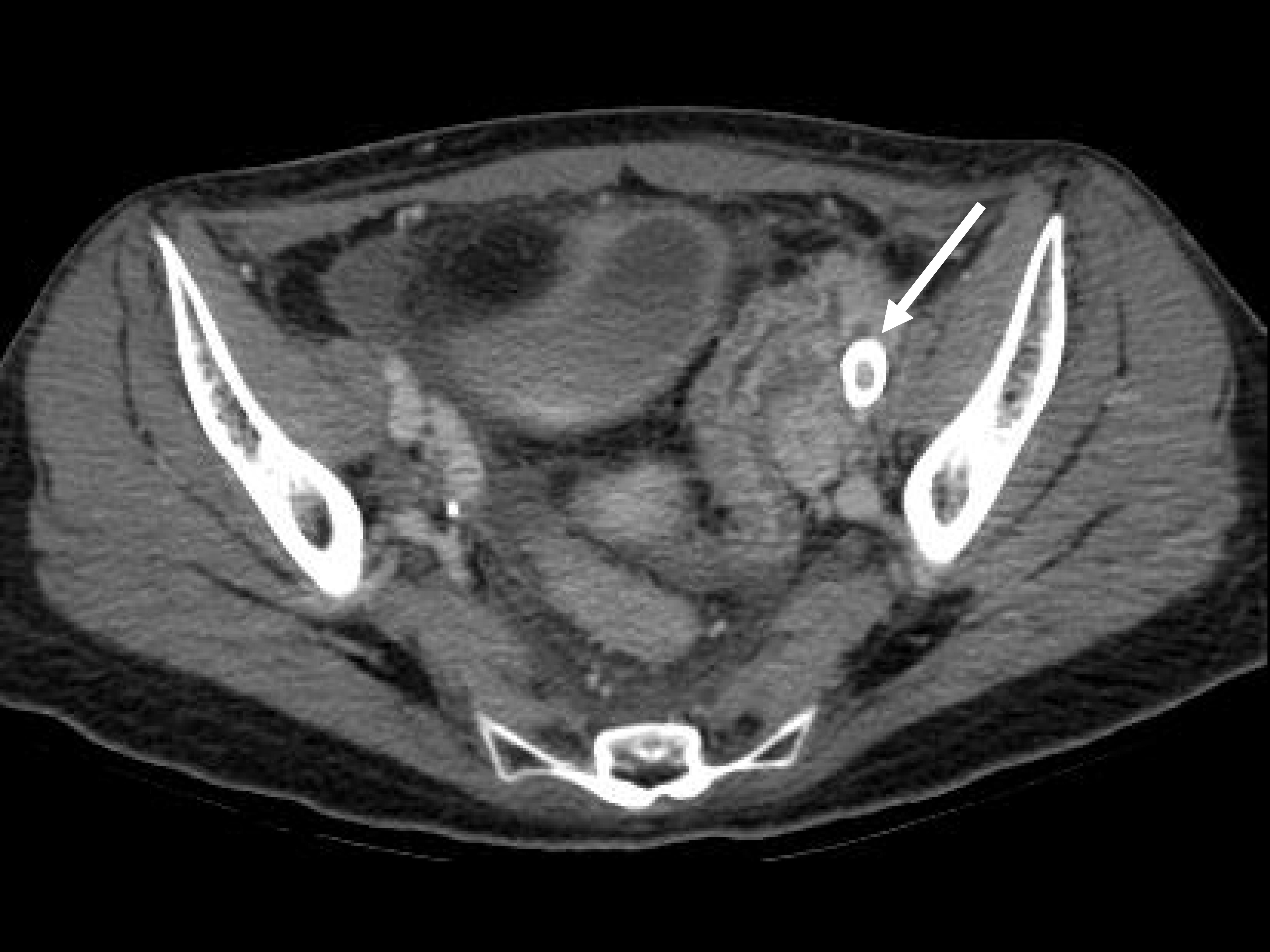
A 38-year-old female patient was admitted to our hospital with sudden hematochezia. She had a history of radical trachelectomy with bilateral pelvic lymph node dissection for stage IB1 cervical cancer and concurrent chemoradiotherapy for recurrence 9 months prior. She was in a stable condition with a heart rate of 72 beats per minute (bpm) and blood pressure of 100/60 mmHg when she visited the emergency room (ER) of our hospital for the first time. Her hemoglobin level was 9.2 g/dL; hematocrit, 25.9%; platelet count, 139,000/µL, and prothrombin time-international normalized ratio (PT-INR), 0.97. Physical examination findings were nonspecific. Sigmoidoscopy revealed no active bleeding. Computed tomography (CT) (Fig. 1A) revealed a thickened wall of the distal ileum and rectosigmoid colon, suggesting radiation-related inflammation. In addition, she no longer complained of hematochezia. Therefore, the patient was discharged.
(A) Computed tomography (CT) image demonstrates thickened wall of the distal ileum and rectosigmoid colon, suggesting radiation-related inflammation, on the first visit. The narrowed left external iliac artery (arrow) contacts air bubbles in the bowel lumen. (B) CT image show the presence of contrast media in almost the entire colon in the delayed phase on the second visit. The left external iliac artery (arrow) is extremely narrow. Considering (A), rupture of the left external iliac artery is retrospectively suspected.
Eight hours later, she revisited the ER because of massive hematochezia. Her hemoglobin level was 9.5 g/dL; hematocrit, 27.2%; platelet count, 101,000/µL; and PT-INR, 1.22. Her blood pressure was 70/40 mmHg, and her vitals stabilized after loading with normal saline. However, she complained of sudden massive hematochezia, and her vitals became unstable, with a blood pressure of 40/20 mmHg and a heart rate of 150 bpm. The patient remained in hypovolemic shock despite massive transfusion. CT was reperformed, which revealed the presence of contrast media in almost the entire colon in the delayed phase (Fig. 1B).
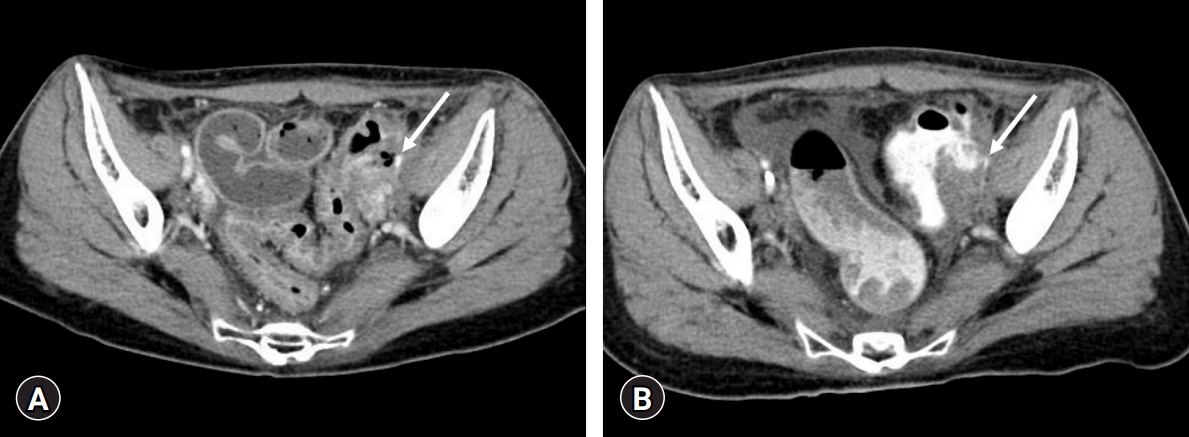
She was referred to the angiography room of the hospital for emergency angiography. Inferior mesenteric arteriography (Fig. 2A) revealed extremely narrow caliber vessels without definite extravasation. Therefore, pelvic arteriography was performed. An arteriogram (Fig. 2B) showed contrast extravasation from the left external iliac artery into the sigmoid colon. This was considered an ilioenteric fistula. A 9 mm × 5 cm Viabahn stent graft (WL Gore and Associates, Flagstaff, AZ, USA) was inserted into the left external iliac artery, followed by ballooning with a 6 mm × 4 cm balloon (Mustang, Boston Scientific, Natick, MA, USA). However, extravasation was still visible, and the patient remained in an unstable condition. Therefore, we inserted another Viabahn stent graft (9 mm in diameter and 10 cm in length) (Fig. 2C). Continuous extravasation was observed despite further ballooning (Fig. 2D). We assumed that the stent graft could not fully expand, and extravasation continued because she was in a hypovolemic state. It was thought that when the patient’s vital signs recovered, the stent graft would attach to the vessel wall. Therefore, despite continuous extravasation, we decided to stop the procedure because her vital signs were stable.

(A) Inferior mesenteric arteriography shows significantly narrow vessels without definite contrast media extravasation. (B) The pelvic arteriography shows extravasation (arrow) from the left external iliac artery into the sigmoid colon. (C) A Viabahn stent graft (5 cm in length; WL Gore and Associates, Flagstaff, AZ, USA) is placed in the left external iliac artery to stop the extravasation, but continuous extravasation is observed despite repeated ballooning. Therefore, another Viabahn stent graft (10 cm in length) is inserted sequentially (arrows at the end of the stent graft). (D) After stent graft placement, ballooning is repeated to put the stent graft and vessel wall together. However, slight extravasation(arrow) is seen on the final angiography. We stopped the procedure because the patient’s condition was stable at that time.
She remained in the intensive care unit for 4 days after the procedure. Her vital signs were stable, and her hemoglobin level stabilized to 16.1 g/dL. Antibiotics and anticoagulants were also administered. She was discharged approximately 1 month later, without any additional bleeding episodes. A month later, she visited the hospital because of swelling of her left leg. Lower extremity CT venography (Fig. 3) showed a patent lumen of the stent graft with no extravasation of contrast media. At that time, we considered the swelling to be lymphedema.
Discussion
A fistula between the major arteries and the gastrointestinal tract is not common but is often life-threatening. Primary arterioenteric fistulas most frequently result from aneurysmal disease but may also be associated with peptic ulcer disease, malignancy, radiation therapy, trauma, diverticulitis, appendicitis, and pancreatic pseudocysts. Secondary fistulas develop after vascular surgery for abdominal aortic aneurysm or aortoiliac occlusive disease and are observed 10 times more often than primary fistulas [2]. Therefore, primary arterioenteric fistulas are extremely rare, with an incidence of 0.07% in the general population [3]. Primary fistulas most commonly occur between the aorta and the esophagus or duodenum and rarely between the iliac artery and the intestine.
Bleeding from a fistula may be minor or intermittent, and there is a symptom-free period before it opens. When a fistula opens, rapid and massive bleeding can be seen [4]. To demonstrate the origin of bleeding from an arterioenteric fistula, CT should be performed while massive bleeding occurs, at which time extravasation of contrast media into the bowel lumen is visible. Therefore, the diagnosis is challenging. According to the literature, CT has varying sensitivity (40%–90%) and specificity (33%–100%) for the diagnosis of arterioenteric fistulas [5].
The standard treatment for an arterioenteric fistula is surgery, including removal of all infected tissue, repair of the bowel defect, and revascularization of the vessel to the lower extremity. It is associated with high morbidity and mortality rates [6,7]. Consequently, less invasive endovascular treatments, such as stent graft exclusion, have been introduced. Antoniou et al. [8] reviewed 33 articles on arterioenteric fistulas published between 1990 and 2008 and found that 41 patients were treated with stent grafts. Complications occurred in 21 patients, of whom 18 developed infections. The authors concluded that stent graft exclusion should not be performed as a final treatment; however, it can be applied as a bridge option to surgical treatment, especially in patients with signs of infection or in emergencies.
Massive gastrointestinal bleeding necessitates emergency angiography, usually celiac, superior mesenteric, and inferior mesenteric arteriographies. However, nonvisceral arteries can be a bleeding focus in postoperative or cancer patients, as in our patient. Therefore, aortography and pelvic arteriography should be performed in such cases [9].
In conclusion, angiography of the aortoiliac arteries is needed when identifiable bleeding sources are not detected on visceral angiography in cases of massive arterial bleeding, especially in postoperative or cancer patients. In addition, endovascular management using a stent graft for arterioenteric fistulas can be a good treatment option for emergencies.
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Funding
This study was supported by a 2019 research grant from Pusan National University Yangsan Hospital.
Author contributions
Conceptualization: JYJ, JHK; Formal analysis: UBJ, JYH; Funding acquisition: JYJ; Resources: JYJ, TUK, HSR; Supervision: UBJ; Writing - original draft: JYJ; Writing - review & editing: all authors.