PubMed Central, CAS, DOAJ, KCI

Articles
- Page Path
- HOME > J Yeungnam Med Sci > Volume 40(Suppl); 2023 > Article
-
Case report
Thyroid storm caused by metastatic papillary thyroid carcinoma tissue after total thyroidectomy: a case report -
So Hee Kwon1
 , Min-Ji Kim1
, Min-Ji Kim1 , Sin Yeong Jung2
, Sin Yeong Jung2 , Jae-Han Jeon1
, Jae-Han Jeon1
-
Journal of Yeungnam Medical Science 2023;40(Suppl):S93-S97.
DOI: https://doi.org/10.12701/jyms.2023.00199
Published online: May 17, 2023
1Department of Internal Medicine, Kyungpook National University Chilgok Hospital, School of Medicine, Kyungpook National University, Daegu, Korea
2Department of Nuclear Medicine, School of Medicine, Kyungpook National University, Daegu, Korea
- Corresponding author: Jae-Han Jeon, MD, PhD Department of Internal Medicine, Kyungpook National University Chilgok Hospital, School of Medicine, Kyungpook National University, 807 Hoguk-ro, Buk-gu, Daegu 41404, Korea Tel: +82-53-200-7201 • Fax: +82-53-200-3155 • E-mail: jeonjh@knu.ac.kr
Copyright © 2023 Yeungnam University College of Medicine, Yeungnam University Institute of Medical Science
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Thyroid storm is a life-threatening form of thyrotoxicosis and an endocrinological emergency. We present a case of thyroid storm in a patient with metastatic papillary thyroid cancer. A 67-year-old woman with a history of total thyroidectomy 4 years prior to presentation was admitted with deteriorating mental status, fever, and tachycardia. Laboratory tests revealed severe thyrotoxicosis. Although the patient had no residual thyroid tissue after total thyroidectomy, she had a previously diagnosed metastatic thyroid cancer lesion in the pelvic bone. Despite initial treatment with a standard thyroid storm regimen, the patient died 6 days after hospitalization. The patient had no history of Graves disease; however, a thyroxine receptor antibody was detected postmortem. The patient had a history of exposure to an iodine contrast agent, which is a rare cause of thyrotoxicosis. Thyroxine production from a differentiated thyroid carcinoma is rare but can be a source of clinically significant thyrotoxicosis in patients post-thyroidectomy. Overlapping Graves disease is a common stimulus; however, other causes, such as exogenous iodine, cannot be excluded. This case demonstrates that in the setting of metastatic thyroid carcinoma, thyrotoxicosis cannot be completely ruled out as a cause of suspicious symptoms, even in patients with a history of total thyroidectomy.
- Differentiated papillary thyroid carcinoma (PTC) cells may possess intact thyroid-stimulating hormone (TSH) receptors and retain the ability to produce thyroxine when stimulated [1]. There are few reports of patients with metastatic PTC after total thyroidectomy who present with thyrotoxicosis that may be caused by antibody-mediated TSH receptor stimulation from Graves disease in the remaining well-differentiated metastatic tissue [2].
- Here, we describe the case of a patient who presented with thyroid storm caused by a metastatic PTC lesion after total thyroidectomy.
Introduction
- Ethical statements: This study was exempt from review by the Institutional Review Board (IRB) of Kyungpook National University Chilgok Hospital (IRB No: 2023-03-008) and the requirement for informed consent from the patient was waived by the IRB.
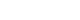
- A 67-year-old woman presented with drowsiness and fever 4 years after total thyroidectomy. In February 2017, the patient, who had a history of type 2 diabetes mellitus and hypertension, was diagnosed with PTC of the thyroid isthmus after a routine health checkup, thyroid ultrasonography (USG), and USG-guided fine-needle aspiration. Preoperative laboratory results showed normal thyroid function, with a serum thyroglobulin level of 102.50 ng/mL (Fig. 1). The patient underwent a total thyroidectomy with central neck lymph node dissection at our institution. Histopathological studies detected a 0.9-cm PTC nodule at the isthmus, along with two benign hyperplastic nodules. No cervical lymph node invasion was detected and no postoperative radioiodine therapy was planned. The patient was prescribed levothyroxine (0.125 mg per day) postoperatively.
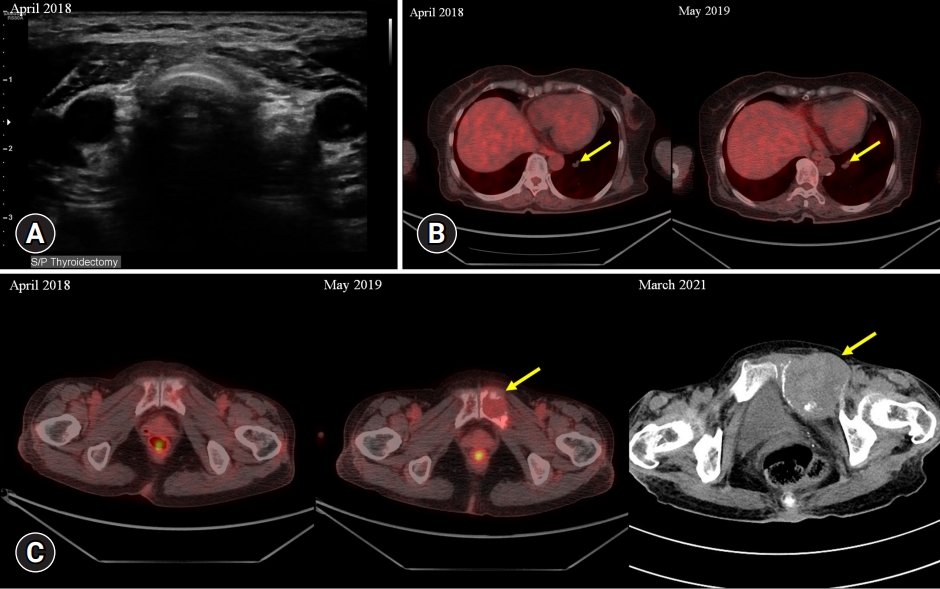
- One year after surgery (April 2018), the patient underwent a follow-up cervical USG, and no visible nodules were found on the thyroid bed (Fig. 2A). Positron emission tomography (PET)/computed tomography (CT) was performed. A 1.3-cm pulmonary nodule was observed without fluorodeoxyglucose (FDG) uptake; at the time of assessment, it was not considered definitive evidence of metastasis (Fig. 2B). The patient’s serum thyroglobulin level was 110.40 ng/mL (Fig. 1). At a subsequent visit (October 2018), the patient remained euthyroid with adequate TSH suppression but complained of weight loss, and the levothyroxine dose was adjusted to 0.1 mg per day.
- After 1 year (May 2019), an increase in serum thyroglobulin (8,820 ng/mL; range, 0–50 ng/mL) was found during a regular follow-up visit, and an additional PET/CT scan was ordered (Fig. 2B). A 4-cm osteolytic mass with FDG uptake was detected in the left pubis. The previously known lung lesion had increased in volume. Radioiodine ablation therapy for metastatic PTC was recommended; however, after consultation, the patient voluntarily decided to forego 131-iodine therapy and received only palliative care. The patient continued follow-up visits to the outpatient clinic of the Department of Nuclear Medicine. The patient was prescribed levothyroxine.
- In November 2020, she visited the outpatient clinic with laboratory results from an external institution obtained in October 2020 that showed marked thyrotoxicosis, and she was advised to decrease the dose of levothyroxine from 0.1 to 0.05 mg daily. After 1 month, laboratory test results showed partially fixed thyrotoxicosis (TSH, 0.02 mIU/L [range, 0.3–4.0 mIU/L]; free thyroxine [T4], 1.16 ng/dL [range, 0.89–1.8 mg/dL]; and triiodothyronine [T3], 2.50 ng/mL [range, 0.6–1.9 ng/mL]), and the attending physician maintained the dose of levothyroxine at 0.05 mg daily.
- In March 2021, she visited our emergency department complaining of abdominal pain and a mass in the lower abdomen. Iodine contrast-enhanced CT showed that the pelvic bone metastasis had increased in size from 4 cm to 6.4 cm (Fig. 2C).
- On April 6, 2021, she visited our emergency department with mental deterioration attributed to hypoglycemia due to glimepiride use at the time of admission. Although the patient’s mental state improved after the intravenous injection of dextrose solution, the initial laboratory workup and chest X-ray showed leukocytosis and lung consolidations, suggesting aspiration pneumonia. At this time, there were no specific symptoms suggestive of thyrotoxicosis, and her thyroid function was not checked. She underwent a contrast-enhanced iodine chest CT scan, which revealed consolidative lesions in the right lung. The patient was prescribed oral antibiotics and was discharged.
- On April 11, 2021, she was found semiconscious and febrile at home with a blood sugar level of 45 mg/dL. She was readmitted to our emergency department. At the time of the initial examination, she had a body temperature of 37.4°C, a heart rate of 117 beats/minute, and a blood pressure of 174/83 mmHg. After the administration of intravenous dextrose, her mental status briefly improved, but her condition deteriorated as her body temperature increased to 38.5°C, with supraventricular tachycardia that was unresponsive to chemical cardioversion with adenosine and verapamil. She received another abdominal and chest CT scan with iodine contrast because her initial vital signs and mild leukocytosis suggested sepsis. However, she showed normal C-reactive protein levels and her thyroid function test showed marked thyrotoxicosis with TSH of <0.008 µIU/mL, free T4 of >12.0 ng/dL, and total T3 of 5.43 ng/mL, indicating a thyroid storm. The patient’s symptoms were summarized using the Burch-Wartofsky Point Scale to confirm the diagnosis of a thyroid storm (Table 1) [3,4]. After excluding accidental or deliberate levothyroxine overdose, the initial suspected diagnosis was a thyroid storm of metastatic thyroid tissue due to undiagnosed Graves disease or other causes of thyrotoxicosis. The patient was initially treated with an oral aqueous potassium iodide solution, 200-mg propylthiouracil three times daily, and propranolol 40 mg four times daily administered via a nasogastric tube because of the patient’s stuporous mental state. Intravenous hydrocortisone (50 mg) was also administered four times daily. Despite initial treatment, the thyrotoxicosis persisted (TSH of <0.01 µIU/mL and free T4 of 5.60 mg/dL on day 5 of hospitalization). There was no improvement in the fever, mental status, or tachycardia.
- On day 5 of hospitalization, the patient developed tachypnea with a Cheyne-Stokes pattern, hypoxia, and hypotension. The patient had previously been on a palliative care plan and refused to undergo invasive resuscitation or life-sustaining invasive management. The patient died on day 6 of hospitalization. An antithyroid antibody test performed 3 days later showed an increased thyrotropin binding inhibitor immunoglobulin titer of 8.86 IU/L (range, 0.0–1.0 IU/L).
- A timeline of the patient’s treatment history and thyroid status is shown in Fig. 1. USG, PET/CT, and CT images of the patient’s disease course are shown in Fig. 2.
Case
- Thyroid storm is an endocrine emergency with a myriad of life-threatening symptoms and multiorgan failure due to a rapid increase in serum thyroid hormone levels. Due to the severity of its symptoms and high mortality [5], this condition requires immediate intervention and management [6]. The pathophysiology of a thyroid storm is not fully understood; however, it is thought to be caused by superimposed insults in patients with existing thyrotoxicosis. A thyroid storm can occur in any patient with hyperthyroidism, including those with hormonally functional thyroid carcinoma [7]. Triggers of thyroid storm include infection, thyroid and non-thyroid surgery, parturition, trauma, stroke, myocardial infarction, hypoglycemia, and exogenous iodine exposure from medications, including amiodarone and contrast agents. The precipitating factor has never been found in 25% to 43% of patients [3]. Patients admitted for thyroid storms are usually treated with measures to decrease excess thyroid activity. Antithyroid agents such as thiamazole are administered as soon as possible in combination with beta-blockers and glucocorticoids, and a large dose of inorganic iodine is recommended along with antithyroid medication (250 mg of saturated potassium iodide every 6 hours, starting 1 hour after the initial dose of antithyroid drug) to decrease the concentration of circulating thyroxine, prevent the organification of iodide (Wolff-Chaikoff effect), and reduce thyroid vascularity [8,9]. In patients who do not respond or are intolerant to antithyroid medications, or if corticosteroids are not effective, the binding agent cholestyramine can be used to physically remove thyroxine from the enterohepatic circulation [10]. In the absence of clinical improvement after 24 to 48 hours, therapeutic plasmapheresis can rapidly remove thyroxine-binding proteins, excess catecholamines, cytokines, and antithyroid receptor antibodies from circulation [11].
- There are a few reports of a thyroid storm caused by hormonally active metastatic thyroid cancer tissue after thyroidectomy, which is more commonly reported in patients with follicular thyroid cancer [2]. A meta-analysis of thyrotoxicosis in patients with thyroid carcinoma caused by hyperfunctioning metastatic carcinoma included 14 patients who presented with thyrotoxicosis originating from distant metastatic tissue. In such cases, medical management with antithyroid medications alone often fails to resolve the thyrotoxicosis, although multidose radioiodine treatment can improve outcomes [6,12]. A metastatic follicular thyroid carcinoma of the pelvic bone that was treated with radioiodine therapy deteriorated into a thyrotoxic lesion with radioiodine resistance; in that case, palliative resection of the lesion resolved the thyrotoxicosis [13].
- In summary, we have described a patient who presented with a thyroid storm with a Burch-Wartofsky Point Scale score of 55. The patient had undergone total thyroidectomy for PTC, and deteriorating metastatic PTC tissue was detected in the bone by PET/CT and confirmed by elevated thyroglobulin levels prior to this presentation. The patient had no known history of Graves disease. However, there are reports of functional thyroid carcinoma tissue as the sole cause of thyrotoxicosis, with or without underlying Graves disease. Several factors can trigger thyroid crisis in the presence of hormonally functional thyroid tissue, including infection, frequent episodes of hypoglycemia, and recent exposure to CT contrast agents. In this case, the patient did not respond to initial treatment with antithyroid medication, inorganic iodide, and corticosteroids. This patient would have had a better chance of survival if further secondary therapies, such as emergency plasmapheresis and eventual radioiodine ablation, had been attempted. However, the patient refused further treatments. Thyroid storm should be considered in patients presenting with a history of total thyroidectomy because autonomous thyroxine production in the remaining metastatic thyroid carcinoma can lead to thyrotoxicosis and even a thyroid storm.
Discussion
-
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
-
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No.2022R1C1C101089811).
-
Author contributions
Conceptualization, Formal analysis: all authors; Data curation: MJK, SYJ, JHJ; Funding acquisition, Project administration, Resources, Supervision, Validation: SYJ, JHJ; Visualization: SHK, MJK; Writing-original draft: SHK, MJK; Writing-review & editing: MJK.
Notes
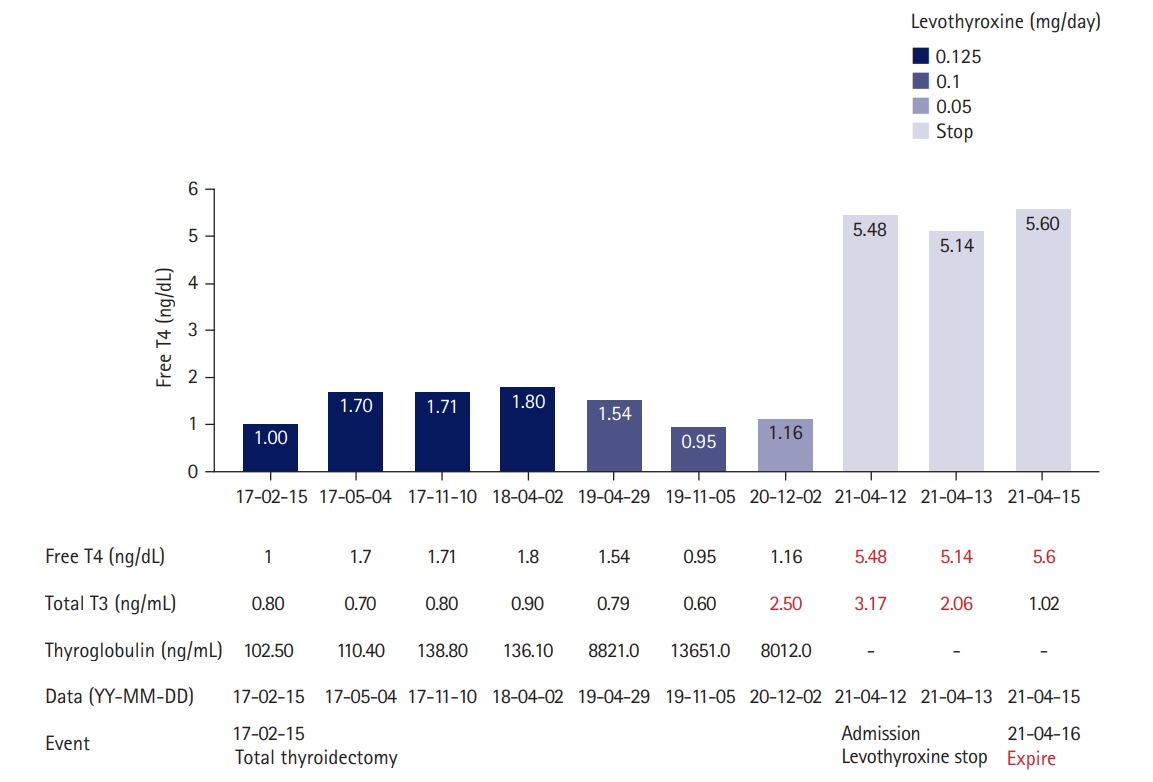

| Variable | Point | In patient (criteria/point) |
|---|---|---|
| Body temperature (˚C) | ||
| 37.2–37.7 | 5 | 38.5/15 |
| 37.8–38.3 | 10 | |
| 38.4–38.8 | 15 | |
| 38.9–39.3 | 20 | |
| 39.4–39.9 | 25 | |
| ≥40.0 | 30 | |
| Heart rate (beats/min) | ||
| 90–109 | 5 | 117/10 |
| 110–119 | 10 | |
| 120–129 | 15 | |
| 130–139 | 20 | |
| ≥140 | 25 | |
| Atrial fibrillation | 10 | Absent/0 |
| Congestive heart failure | ||
| None | 0 | None/0 |
| Mild | 5 | |
| Moderate | 10 | |
| Severe | 15 | |
| Gastrointestinal/hepatic effects | ||
| None | 0 | None/0 |
| Moderatea) | 10 | |
| Severeb) | 20 | |
| Central nervous system effects | ||
| Absent | 0 | Moderate/20 |
| Mildc) | 10 | |
| Moderated) | 20 | |
| Severee) | 30 | |
| Precipitating event | 10 | Yes/10 |
| Total score | ||
| ≥45 | Thyroid storm | Storm/55 |
| 25–44 | Impending storm | |
| <25 | Unlikely |
- 1. Ishihara T, Ikekubo K, Shimodahira M, Iwakura T, Kobayashi M, Hino M, et al. A case of TSH receptor antibody-positive hyperthyroidism with functioning metastases of thyroid carcinoma. Endocr J 2002;49:241–5.ArticlePubMed
- 2. Folkestad L, Brandt F, Brix T, Vogsen M, Bastholt L, Grupe P, et al. Total thyroidectomy for thyroid cancer followed by thyroid storm due to thyrotropin receptor antibody stimulation of metastatic thyroid tissue. Eur Thyroid J 2017;6:276–80.ArticlePubMedPMCPDF
- 3. Chiha M, Samarasinghe S, Kabaker AS. Thyroid storm: an updated review. J Intensive Care Med 2015;30:131–40.ArticlePubMedPDF
- 4. Burch HB, Wartofsky L. Life-threatening thyrotoxicosis: thyroid storm. Endocrinol Metab Clin North Am 1993;22:263–77.ArticlePubMed
- 5. Akamizu T, Satoh T, Isozaki O, Suzuki A, Wakino S, Iburi T, et al. Diagnostic criteria, clinical features, and incidence of thyroid storm based on nationwide surveys. Thyroid 2012;22:661–79.ArticlePubMedPMC
- 6. Basida B, Zalavadiya N, Ismail R, Krayem H. Weathering the storm: thyroid storm precipitated by radioiodine contrast in metastatic thyroid carcinoma. Cureus 2021;13:e14219.ArticlePubMedPMC
- 7. Naito Y, Sone T, Kataoka K, Sawada M, Yamazaki K. Thyroid storm due to functioning metastatic thyroid carcinoma in a burn patient. Anesthesiology 1997;87:433–5.ArticlePubMedPDF
- 8. Suwansaksri N, Preechasuk L, Kunavisarut T. Nonthionamide drugs for the treatment of hyperthyroidism: from present to future. Int J Endocrinol 2018;2018:5794054.ArticlePubMedPMCPDF
- 9. Ross DS, Burch HB, Cooper DS, Greenlee MC, Laurberg P, Maia AL, et al. 2016 American Thyroid Association guidelines for diagnosis and management of hyperthyroidism and other causes of thyrotoxicosis. Thyroid 2016;26:1343–421.ArticlePubMed
- 10. Solomon BL, Wartofsky L, Burman KD. Adjunctive cholestyramine therapy for thyrotoxicosis. Clin Endocrinol (Oxf) 1993;38:39–43.ArticlePubMed
- 11. Satoh T, Isozaki O, Suzuki A, Wakino S, Iburi T, Tsuboi K, et al. 2016 Guidelines for the management of thyroid storm from The Japan Thyroid Association and Japan Endocrine Society (first edition). Endocr J 2016;63:1025–64.ArticlePubMed
- 12. Liu J, Wang Y, Da D, Zheng M. Hyperfunctioning thyroid carcinoma: a systematic review. Mol Clin Oncol 2019;11:535–50.ArticlePubMedPMC
- 13. Tan J, Zhang G, Xu W, Meng Z, Dong F, Zhang F, et al. Thyrotoxicosis due to functioning metastatic follicular thyroid carcinoma after twelve I-131 therapies. Clin Nucl Med 2009;34:615–9.ArticlePubMed
References
Figure & Data
References
Citations

- Data Analysis and Systematic Scoping Review on the Pathogenesis and Modalities of Treatment of Thyroid Storm Complicated with Myocardial Involvement and Shock
Eman Elmenyar, Sarah Aoun, Zain Al Saadi, Ahmed Barkumi, Basar Cander, Hassan Al-Thani, Ayman El-Menyar
Diagnostics.2023; 13(19): 3028. CrossRef

 E-Submission
E-Submission Yeungnam University College of Medicine
Yeungnam University College of Medicine PubReader
PubReader ePub Link
ePub Link Cite
Cite