PubMed Central, CAS, DOAJ, KCI

Articles
- Page Path
- HOME > J Yeungnam Med Sci > Volume 40(Suppl); 2023 > Article
-
Original article
Association between total body muscle percentage and prevalence of non-alcoholic fatty liver disease in Korean adults findings from an 18-year follow-up: a prospective cohort study -
Byoung Chan Ahn1
 , Chul Yong Park2,3
, Chul Yong Park2,3 , Jung Hee Hong4
, Jung Hee Hong4 , Ki Ook Baek2
, Ki Ook Baek2
-
Journal of Yeungnam Medical Science 2023;40(Suppl):S47-S55.
DOI: https://doi.org/10.12701/jyms.2023.00605
Published online: August 29, 2023
1Division of Gastroenterology, Department of Internal Medicine, Keimyung University Dongsan Medical Center, Daegu, Korea
2Department of Occupational and Environmental Medicine, Yeungnam University Hospital, Daegu, Korea
3Department of Preventive Medicine and Public Health, Yeungnam University College of Medicine, Daegu, Korea
4Department of Radiology, Keimyung University Dongsan Medical Center, Daegu, Korea
- Corresponding author: Ki Ook Baek, MD Department of Occupational and Environmental Medicine, Yeungnam University Hospital, 170 Hyeonchung-ro, Nam-gu, Daegu 42415, Korea Tel: +82-10-2663-5408 • Fax: +82-53-653-2061 • E-mail: bko8899@gmail.com
Copyright © 2023 Yeungnam University College of Medicine, Yeungnam University Institute of Medical Science
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 1,717 Views
- 51 Download
Abstract
-
Background
- This study aimed to elucidate the association between total lean muscle mass and the incidence of non-alcoholic fatty liver disease (NAFLD) in the adult Korean population.
-
Methods
- Utilizing data derived from the 18-year prospective cohort of the Korean Genome and Epidemiology Study, NAFLD was diagnosed via the hepatic steatosis index with an established cutoff value of 36. Lean muscle mass was assessed via bioelectrical impedance analysis and subsequently divided into tertiles. A generalized mixed model with a logit link was employed for repeated measures data analysis, accounting for potential confounders.
-
Results
- Analysis encompassed 7,794 participants yielding 49,177 measurements. The findings revealed a markedly increased incidence of NAFLD in the lower tertiles of muscle mass, specifically, tertile 1 (odds ratio [OR], 20.65; 95% confidence interval [CI], 9.66–44.11) and tertile 2 (OR, 4.57; 95% CI, 2.11–9.91), in comparison to tertile 3. Age-dependent decreases in the OR were observed within the tertile 1 group, with ORs of 10.12 at age of 40 years and 4.96 at age of 80 years. Moreover, each 1%-point increment in total muscle mass corresponded with an estimated OR of 0.87 (95% CI, 0.82–0.93) for NAFLD resolution.
-
Conclusions
- The study demonstrates a significant association between total muscle mass and NAFLD prevalence among Korean adults. Given the potential endocrine role of muscle mass in NAFLD pathogenesis, interventions aimed at enhancing muscle mass might serve as an effective public health strategy for mitigating NAFLD prevalence.
- Non-alcoholic fatty liver disease (NAFLD) comprises a spectrum of liver conditions occurring in the absence of significant alcohol consumption. These conditions range from non-alcoholic fatty liver to non-alcoholic steatohepatitis (NASH) and NASH cirrhosis, all typified by excessive hepatic fat accumulation [1]. The global prevalence rate of NAFLD, the most common chronic liver disease, is a striking 32% among adults. In the Republic of Korea, the prevalence has risen sharply, largely attributable to westernized dietary habits, diminished physical activity, and the ensuing increase in obesity and diabetes rates. Further, NAFLD has been identified as an independent risk factor for numerous systemic diseases, including cardiovascular disease, diabetes mellitus, metabolic syndrome, chronic kidney disease, and various malignancies [2-4], thereby elevating its public health and socioeconomic impact.
- The role of skeletal muscle extends beyond locomotion, serving as a secretory organ that produces myokines, substances influencing other organs in an autocrine, paracrine, or endocrine hormone-like manner [5,6]. Specific myokines, such as interleukin 6 (IL-6), secreted from skeletal muscle, modulate the development of NAFLD by acting as endocrine organs that regulate various metabolic processes, including glucose metabolism in the liver or pancreas and lipolysis in adipose tissue [7].
- Previous research has established a correlation between muscle mass and NAFLD, with a majority of such studies being cross-sectional [8]. Nevertheless, there is a growing call for longitudinal research to map this association over time, thereby yielding a more holistic understanding of the muscle mass-NAFLD interplay. Longitudinal studies are crucial for elucidating the dynamic nature of this relationship, which holds significant implications for NAFLD prevention and management. Therefore, our study aimed to examine the association between muscle mass and NAFLD using a Korean population cohort dataset.
Introduction
- Ethical statements: Prior to the survey, all participants were informed that they had been randomly chosen to participate in the Korean Genome and Epidemiology Study (KoGES) survey with the right to refuse to be involved in further analyses, and signed informed consents were obtained. We received the data in fully anonymized form. This study was conducted after obtaining an approval from the Institutional Review Board (IRB) of Yeungnam University Hospital, (IRB No: 2023-05-057). The study was carried out in accordance with the ethical standards of the Helsinki Declaration.
- 1. Study participants
- This study utilized a subset of data derived from the Korean Genome and Epidemiology Study (KoGES), an ongoing initiative by the National Institute of Health (NIH) and the Korea Disease Control and Prevention Agency since 2001 [9]. Permission was secured to access and scrutinize laboratory results and participant questionnaires from the KoGES cohort. The dataset under analysis encompassed a subset of the KoGES cohort consisting of adults aged 40 to 70 years at baseline, residing in the South Korean cities of Ansan and Anseong. Biennial data collection commenced in 2001, with this study utilizing data from the ninth follow-up survey conducted during 2017 to 2018. It should be noted that participant attendance varied per examination phase, with an absence of data explaining non-participation in each phase or exclusion from specific assessments.
- Initial participant selection for this study entailed excluding individuals who consumed over 20 g of alcohol per day, given that the NAFLD diagnosis was confined to non-drinkers or those consuming less than 20 g of alcohol daily [10]. Those with a prior cancer diagnosis were likewise excluded. Further exclusions comprised cases with missing data among the primary confounding variables considered in this study, including the variables instrumental to the operational definition of NAFLD and those absent from the bioelectrical impedance analysis (BIA) of body composition at each measurement time point.
- 2. Sample collection and laboratory analysis
- Laboratory analysis results were procured and utilized from the NIH. Blood samples from the KoGES cohort were drawn from participants and collected in a serum separator tube and two ethylenediaminetetraacetic acid tubes. To optimize long-term storage, both serum and plasma fractions were prepared and divided into six to 10 vials, each containing 300 to 500 μL. All samples were then transported to the National Biobank of Korea for meticulous storage, ensuring their availability for future research applications. Venous blood samples were obtained from study participants after a fasting period of ≥12 hours. Fasting glucose levels were assessed using a glucose oxidase-based assay. Aspartate aminotransferase (AST) and alanine aminotransferase (ALT) activities were quantified using enzymatic methods (Advia 1650 system; Siemens, Tarrytown, NY, USA). For samples procured prior to September 2002, analyses of glucose, AST, and ALT were performed with a HITACHI 7600 (Hitachi, Tokyo, Japan), with data adjusted to account for systematic errors due to equipment transitions.
- 3. Assessment of body composition
- Total muscle mass was assessed using multifrequency BIA (MF-BIA, InBody 3.0; Biospace, Seoul, Korea). Unlike traditional BIA methods, which often provide partial measurements and rely on estimations for whole body composition, MF-BIA posits that the human body comprises five interconnected cylinders, enabling direct impedance measurements from these bodily compartments. An eight-point tetrapolar tactile electrode system was employed to measure impedance at four specific frequencies (5, 50, 250, and 500 kHz) across five segments (right arm, left arm, trunk, right leg, and left leg). These readings were utilized to calculate segmental lean body mass by assessing the intracellular and extracellular water components of total body water. The total muscle weight (in kg) was then divided by the total body weight (in kg) to yield the total body muscle percentage (TBM%).
- 4. Survey and anthropometric assessment
- In this study, information regarding smoking habits, medical history (including diabetes mellitus and cancer), and alcohol consumption was collected through structured questionnaires. The survey incorporated a categorization of smoking status into “current nonsmoker,” “former smoker,” and “current smoker.” For alcohol intake, the questionnaire inquired about the frequency of alcohol use and daily consumption. This latter measure was determined considering the volume and type of alcoholic beverages consumed, their respective alcohol content, and the specific gravity of ethanol. Anthropometric measurements, such as height and weight, were recorded by trained personnel, which enabled the computation of body mass index (BMI; kg/m2) using the formula: weight (kg) divided by the square of height (m).
- 5. Non-alcoholic fatty liver disease categorization based on laboratory data
- NAFLD was diagnosed using the hepatic steatosis index (HSI), a diagnostic tool developed and validated specifically for the Korean population [11]. A standard cutoff value of 36 was applied in the HSI formula:
- In this formula, diabetes was defined either by a self-reported diagnosis in the survey or by fasting glucose levels equal to or exceeding 126 mg/dL in the corresponding year. Participants in survey phases 2 and 3 were questioned, “Have you been diagnosed with diabetes since the previous examination?” Thus, the classification of individuals with diabetes necessitated consideration of responses from each phase and previous examinations. Participants consuming over 20 g of alcohol per day were excluded at the participant selection stage; hence those exceeding the cutoff value were classified as having NAFLD.
- 6. Statistical analysis
- Initially, participant characteristics at each time point were delineated based on the presence or absence of NAFLD, focusing specifically on relevant and confounding variables. Preliminary analysis revealed a significant discrepancy in body weight between NAFLD and non-NAFLD groups, necessitating the use of a weight-adjusted indicator for data analysis. TBM% was categorized by computing the tertile cutoff for each sex, derived from baseline measurements. Consequently, muscle mass at every time point was categorized into tertiles using these cutoff values.
- Given the longitudinal nature of the study, repeated measures, and potential missing data, along with the dynamic variability of NAFLD prevalence and TBM% over time, a generalized linear mixed model was applied for multivariable analysis. This model selection ensured the accommodation of potential non-independence of measurements within individuals. With the outcome variable being binary, representing NAFLD presence or absence, a logit link function was employed. Random effects were incorporated for individuals to account for individual-specific repeated measurements, including random interactions. An adaptive Gauss-Hermite quadrature approximation was not utilized for model estimation. Additionally, an interaction term was included between TBM% and age to explore the variance in the relationship between TBM% and NAFLD according to age. Consideration of potential confounding variables [12]—age, sex, smoking status (nonsmoker, former smoker, current smoker), diabetes mellitus (either self-reported or fasting glucose, ≥126 mg/dL), and obesity (BMI, ≥25 kg/m2)—was applied, as these could possibly influence both the independent and dependent variables and are not in the causal pathway between them.
- To further explore the potential impact of muscle development on the resolution of NAFLD, given the longitudinal nature of the study, we sought to determine the odds ratio (OR) for NAFLD in subsequent evaluations among individuals initially diagnosed with NAFLD, based on the change in TBM% from baseline. A model was constructed using the discrepancy between baseline and follow-up TBM% as an independent variable, excluding the initial data. Generally, a declining trend in the number of participants was observed in tertile 3 over each assessment period, potentially indicative of aging effects seen in longitudinally tracked subjects over an extended timeframe, using the baseline as the reference point. To adjust for this effect, age at each evaluation, rather than baseline age, was accounted for as a confounding variable in the mixed-effects model. While not the primary focus of the investigation, the results for total fat mass proportion (total fat/weight, %), total muscle adjusted by height (total muscle/height2), total muscle mass (kg), and waist circumference (cm) were also reported, utilizing the same statistical methodology. The results were presented as ORs, with statistical significance established at p<0.05.
- All statistical analysis was performed using R software version 4.3.0 (R Foundation for Statistical Computing, Vienna, Austria; https://r-project.org), incorporating the lme4 package for the generalized linear mixed model analysis.
Methods
- 1. General characteristics of participants
- From an initial pool of 10,030 participants in the KoGES cohort at baseline, the study excluded 1,567 individuals identified as high-risk drinkers (consuming >20 g/day of alcohol), 237 individuals with a history of cancer, and 432 individuals with incomplete data relevant to the study. Consequently, the study encompassed a total of 7,794 individuals. Due to attrition, the number of measured cases varied over time: 6,607 in phase 2, 5,796 in phase 3, 5,224 in phase 4, 5,229 in phase 5, 4,899 in phase 6, 4,608 in phase 7, 4,627 in phase 8, and 4,393 in phase 9 (Fig. 1).
- Table 1 provides the cohort’s baseline characteristics. At baseline, the NAFLD and non-NAFLD groups exhibited no significant age difference, with mean ages of 52.7±9.1 and 52.5±8.5 years, respectively. A higher female representation was observed in the NAFLD group, comprising 1,349 individuals (67.3%), compared to 3,433 individuals (59.3%) in the non-NAFLD group. Notably, the prevalence of obesity significantly varied across groups, with 1,550 individuals (26.8%) in the non-NAFLD group versus 1,821 (90.9%) in the NAFLD group.
- For the purposes of categorizing TBM% into three groups, we established cutoff points based on the baseline (2001–2002) tertiles of TBM% in the study’s participants, separated by sex. The tertile cutoff values for males were 72.1% and 76.4%, while for females, they were 62.2% and 66.0%. Among the NAFLD group, the respective proportions for each tertile were 1,373 (68.5%), 500 (25.0%), and 130 (6.5%), which were significantly different from those of the non-NAFLD group, with values of 1,223 (21.1%), 2,092 (36.1%), and 2,476 (42.8%).
- The NAFLD group demonstrated higher AST and ALT levels, with averages of 31.1±22.4 U/L and 39.0±45.8 U/L, respectively, compared to 27.8±15.8 U/L and 22.7±14.8 U/L respectively in the non-NAFLD group. Detailed results and statistical metrics for variables in relation to NAFLD status at each time point, including TBM% changes (% point), are provided in Supplementary Table 1.
- 2. Association between total body muscle percentage and non-alcoholic fatty liver disease
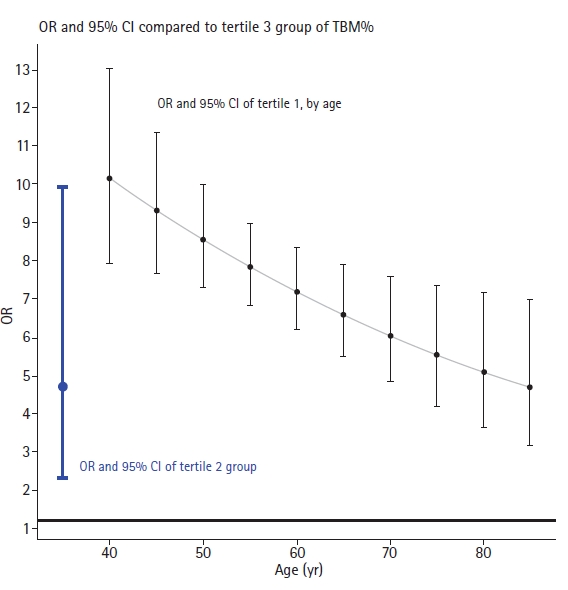
- The results from the generalized linear mixed model analysis revealed significant correlations between TBM% and NAFLD presence when categorized based on tertile cutoffs. Relative to individuals in tertile 3, those in tertile 1 demonstrated an OR for NAFLD of 20.65 (95% CI, 9.66–44.11), suggesting a substantial risk elevation. Similarly, the OR was 4.57 (95% CI, 2.11–9.91) for individuals in tertile 2 compared to tertile 3, implying another significant risk increase. Concerning age, NAFLD saw a significant annual decrease by 0.97 (95% CI, 0.95–0.98). The interaction between TBM% in tertile 1 and age yielded a statistically significant result of 0.98 (95% CI, 0.97–1.00; p<0.01). However, the interaction between age and tertile 2 was not statistically significant (p=0.06) (Table 2). Accounting for these interaction terms, individuals in tertile 1 had an OR for NAFLD of 10.12 (95% CI, 7.85–13.05) at age of 40 years, 8.47 (95% CI, 7.21–9.95) at age of 50 years, 7.09 (95% CI, 6.72–8.92) at age of 60 years, 5.93 (95% CI, 4.68–7.51) at age of 70 years, and 4.96 (95% CI, 3.49–7.06) at age of 80 years, relative to those in tertile 3 (Fig. 2).
- In the model examining the influence of TBM% alterations on NAFLD resolution among initial NAFLD cases, a 1%-point increase in TBM% corresponded to an OR of 0.87 (95% CI, 0.82–0.93) for NAFLD. Yet, the interaction between TBM% at different time points was not statistically significant (Table 3).
- Models incorporating other body composition indices are provided exclusively Supplementary Table 2, presenting the adjusted ORs for each index. Total muscle mass, total muscle adjusted by height, and fat proportion exhibited positive associations with NAFLD prevalence.
Results
- In this study, we scrutinized 18 years’ worth of cohort data to discern the relationship between TBM% and NAFLD prevalence. Individuals grouped within the highest tertile (tertile 3) of TBM% presented with notably reduced NAFLD prevalence when contrasted with those in tertiles 1 and 2. The effect size, represented by the discrepancy in NAFLD prevalence between tertiles 1 and 3, exhibited a diminishing trend as age increased. Yet, even at the advanced age of 80 years, the effect size remained substantial and statistically significant with an OR of 4.96. Conversely, the NAFLD prevalence difference between tertile 2 and tertile 3 did not demonstrate notable age-related variations. With an OR of 4.57, the effect size for the second tertile was generally lower relative to tertile 3, thereby suggesting a dose-dependent reduction in NAFLD risk with increased muscle mass, notwithstanding the influence of aging. Furthermore, the enhancement of muscle mass among individuals with NAFLD was correlated with NAFLD resolution, irrespective of TBM%. However, when assessing factors such as total muscle mass and muscle mass adjusted by height—factors that do not account for body weight—conflicting findings emerged. These inconsistencies might be attributed to the substantial weight differential, with the NAFLD group displaying approximately 10 kg more weight compared to their non-NAFLD counterparts. Therefore, it appears appropriate to employ the weight-adjusted indicator TBM% in such analyses.
- Skeletal muscle, often perceived as an endocrine and paracrine organ, secretes myokines which exert effects on various organs and systems, including the liver, adipose tissue, pancreas, bones, and cardiovascular system. Moreover, skeletal muscle, serving as a primary organ responsible for insulin-mediated glucose uptake, has a close association with insulin resistance [13]. The condition of reduced skeletal muscle mass, known as sarcopenia, has been linked to metabolic syndrome [14], cardiovascular disease [15], diabetes mellitus [16], and even increased overall mortality [17], as reported in several systematic review studies.
- Sarcopenia shares several risk factors with NAFLD and other metabolic disorders, such as obesity and metabolic syndrome, albeit via distinct mechanisms. Pathways involving systemic inflammation and insulin resistance are implicated in both disorders, with a decrease in skeletal muscle mass contributing to decreased insulin sensitivity and energy expenditure, thereby promoting increased hepatic gluconeogenesis, elevated hepatic free fatty acid uptake, and reduced fatty acid oxidation [18]. Moreover, the suppression of muscle peroxisome proliferator-activated receptor γ coactivator-1 α [19], IL-6, irisin [20], along with other molecules secreted by skeletal muscle, is associated with heightened risk of metabolic disorders and NAFLD [21]. However, potential pathways through which NAFLD impacts skeletal muscle have been proposed [22], and in epidemiological studies seeking to ascertain causality direction, a clear consensus remains elusive [23]. Some schools of thought suggest an association rooted in a common etiological basis [24].
- Epidemiological research has thoroughly investigated the correlation between muscle mass and NAFLD or NASH. Past studies have examined the association between NAFLD or NASH and reduced muscle mass within the context of sarcopenia as well as reported the relationship between skeletal mass index and NAFLD prevalence. A 2020 systematic review, incorporating 19 studies for meta-analysis, demonstrated significantly lower skeletal mass index in NAFLD patients and a significant relationship between sarcopenia and increased prevalence of NAFLD, NASH, and NAFLD-related fibrosis [8]. Although the majority of studies examining the NAFLD and sarcopenia relationship, including those in the systematic review, were of cross-sectional design, some longitudinal studies have also been reported. Notably, Kim et al. [25] analyzed a cohort dataset of 12,624 individuals recruited from a medical institution and found the highest tertile of skeletal muscle mass to significantly decrease future NAFLD incidence (adjusted hazard ratio [aHR], 0.44; 95% CI, 0.38–0.51) in the no-NAFLD baseline group, while increasing the future NAFLD resolution rate in the NAFLD baseline group (aHR, 2.09; 95% CI, 1.02–4.28). In a separate study, Lee et al. [26] analyzed a cohort of individuals undergoing health screenings at a different hospital, identified the presence or absence of NAFLD via ultrasound, and discovered that individuals without NAFLD had an increased risk of developing NAFLD over a 10-year period based on muscle and adipose tissue mass levels. The study revealed that higher appendicular skeletal muscle mass in non-obese individuals was associated with a lower prevalence of NAFLD (OR, 1.38; 95% CI, 1.34–2.45).
- Our study, in contrast to prior research, holds the advantage of sampling a broad population rather than a patient group from medical facilities, thus mitigating selection bias. Moreover, we leveraged an expansive dataset collected over an 18-year duration. The use of mixed models enabled the inclusion of individuals diagnosed with NAFLD at baseline while accommodating the dynamic fluctuations in NAFLD and muscle mass throughout the follow-up period. The study also considered age-associated changes in the relationship. As age increases, the diminishing OR of NAFLD in relation to TBM% could be ascribed to the increased influence of age-correlated NAFLD risk factors such as dyslipidemia [27] and altered thyroid function [28]. These conditions may exert a heightened impact on NAFLD development among the elderly, thereby weakening the correlation between TBM% and NAFLD. Nevertheless, it is significant that a noteworthy OR was observed even among older subjects. This finding reinforces that the relationship between TBM% and NAFLD remains robust within the aging population, suggesting that muscle mass continues to play a pivotal role in NAFLD pathogenesis, notwithstanding the amplified effect of age-related risk factors.
- Additionally, our findings underscore that temporal alterations in muscle mass could modulate NAFLD resolution. This constitutes a strength of our study, given its longitudinal structure and the model’s capacity to account for changes in both TBM% and NAFLD status. Regardless of TBM% per se, shifts in muscle mass were linked to a decrease in NAFLD prevalence. In light of previously understood mechanisms, it can be inferred that an individual-level increase in muscle mass may lessen the NAFLD risk, thereby suggesting the potential efficacy of public health initiatives aimed at augmenting muscle mass for NAFLD reduction.
- A limitation of this study lies in the diagnostic modality employed for NAFLD categorization. While we utilized the HSI index, a validated tool for NAFLD diagnosis among Koreans, we did not use direct diagnostic tools such as ultrasound. Additionally, the usage of a secondary dataset not primarily intended for NAFLD research could introduce potential misclassification, as it may not definitively exclude hepatic conditions resulting from medication use, viral infection, autoimmune disease, among others. Nevertheless, given the availability of data on alcohol consumption, we adopted a more stringent criterion to minimize misclassification. Diverse criteria have been applied to distinguish NAFLD from alcoholic fatty liver disease based on alcohol consumption, with some studies implementing thresholds of 30 g/day [29] or 40 g/day [30]. In our study, we opted for a relatively strict threshold of 20 g/day [10], aiming to enhance the specificity of the operational definition of NAFLD at the cost of potentially decreased sensitivity. Another limitation pertains to our usage of TBM% as a surrogate measure, instead of skeletal muscle, which bears clinical significance for the diagnosis of sarcopenia. Furthermore, muscle mass measurements were not obtained through techniques such as dual-energy X-ray absorptiometry, computed tomography, or magnetic resonance imaging, and the lack of control over factors like hydration in muscle measurement using BIA presents another limitation. However, it is noteworthy that TBM% estimated using BIA is known to correlate well with dual-energy X-ray absorptiometry results [31]. In order to mitigate potential overestimation issues with BIA, our study analysis was based on tertiles of muscle mass rather than muscle weight or proportion, thereby reducing the potential misclassification of participants due to differences in muscle measurement methods. Therefore, the impact of misclassification based on muscle measurement methods is likely minimal in our analysis. Another limitation to consider is that while the study elucidates the association between NAFLD and TBM%, it does not establish a causal relationship in which muscle mass precedes NAFLD.
- Notwithstanding its limitations, this study underscores that TBM% is an important independent factor associated with NAFLD among the Korean adult demographic. This highlights the integral role muscle tissue plays in managing NAFLD within this population. Considering the presumptive causality suggested by muscle in light of previous mechanistic investigations, alongside the association of muscle mass alterations with NAFLD resolution, interventions directed toward muscle mass enhancement could substantially impact NAFLD reduction. Owing to the relative ease of quantifying muscle mass via BIA devices, this factor can be leveraged in the realm of public health to manage NAFLD, through venues such as nationwide health examinations or health promotion initiatives.
Discussion
-
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
-
Acknowledgments
Data in this study were from the Korean Genome and Epidemiology Study (KoGES; 4851-302), National Institute of Health, Korea Disease Control and Prevention Agency, Republic of Korea.
-
Funding
None.
-
Author contributions
Conceptualization: BCA, JHH, KOB; Data curation: CYP; Formal analysis: JHH, KOB; Methodology: CYP, KOB; Project administration, Supervision: KOB; Visualization, Resources: JHH; Investigation: BCA; Writing-original draft: BCA; Writing-review & editing: CYP, JHH, KOB.
Notes
Supplementary materials
Supplementary Table 1.
Supplementary Table 2.

| Characteristic | Phase 1 (baseline, 2001–2002) (n=7,794) | ||
|---|---|---|---|
| No NAFLD (HSI ≤36) | NAFLD (HSI >36) | p-valuea) | |
| No. of paients | 5791 | 2003 | |
| Age (yr) | 52.7±9.1 | 52.5±8.5 | 0.506 |
| Weight (kg) | 58.9±9.0 | 69.5±9.1 | <0.001 |
| Body mass index (kg/m2) | 23.4±2.4 | 28.0±2.5 | <0.001 |
| <25 | 4,241 (73.2) | 182 (9.1) | |
| ≥25 | 1,550 (26.8) | 1,821 (90.9) | |
| Category of TBM%b) | <0.001 | ||
| Tertile 1 | 1,223 (21.1) | 1,373 (68.5) | |
| Tertile 2 | 2,092 (36.1) | 500 (25.0) | |
| Tertile 3 | 2,476 (42.8) | 130 (6.5) | |
| Sex | <0.001 | ||
| Male | 2,358 (40.7) | 654 (32.7) | |
| Female | 3,433 (59.3) | 1,349 (67.3) | |
| Smoking | <0.001 | ||
| Nonsmoker | 3,805 (65.7) | 1,430 (71.4) | |
| Past smoker | 806 (13.9) | 237 (11.8) | |
| Current smoker | 1,180 (20.4) | 336 (16.8) | |
| Diabetes mellitusc) | <0.001 | ||
| No | 5,512 (95.2) | 1,644 (82.1) | |
| Yes | 279 (4.8) | 359 (17.9) | |
| AST (U/L) | 27.8±15.8 | 31.1±22.4 | <0.001 |
| ALT (U/L) | 22.7±14.8 | 39.0±45.8 | <0.001 |
Values are presented as number only, mean±standard deviation, or number (%).
NAFLD, non-alcoholic fatty liver disease; HSI, hepatic steatosis index. TBM%, total body muscle percentage; AST, aspartate aminotransferase; ALT, alanine aminotransferase.
a) The p-value was calculated by t-test in continuous variables, and chi-square test in categorical variables.
b) Tertile of TBM% was determined by baseline data of participants. Cutoff for tertile is 72.1% and 76.4% for male, and 62.2% and 66.0% for female.
c) Diagnosed by physician or fasting glucose of ≥126 mg/dL.
| Variable | OR (95% CI)a) | p-value |
|---|---|---|
| Category of TBM% | ||
| Tertile 3 | Reference | |
| Tertile 1 | 20.65 (9.66–44.11) | <0.001 |
| Tertile 2 | 4.57 (2.11–9.91) | <0.001 |
| Age (yr) | 0.97 (0.95–0.98) | <0.001 |
| Obesity | ||
| No (BMI <25 kg/cm2) | Reference | |
| Yes (BMI ≥25 kg/cm2) | 15.27 (13.64–17.1) | <0.001 |
| Sex | ||
| Male | Reference | |
| Female | 2.1 (1.78–2.47) | <0.001 |
| Diabetes mellitusb) | ||
| No | Reference | |
| Yes | 10.48 (8.64–12.72) | <0.001 |
| Smoking | ||
| Nonsmoker | Reference | |
| Past smoker | 1.05 (0.89–1.25) | 0.547 |
| Current smoker | 1.26 (1.04–1.51) | <0.050 |
| Interaction term | ||
| Age× tertile 1 of total muscle mass | 0.98 (0.97–1.00) | <0.010 |
| Age×tertile 2 of total muscle mass | 0.99 (0.97–1.00) | 0.060 |
OR, odds ratio; CI, confidence interval; TBM%, total body muscle percentage; BMI, body mass index.
a) OR, 95% CI, and p-values were calculated using generalized linear mixed model with logit link function. Each participant was regarded as random effect, and other suggested variables were regarded as fixed effect.
b) Diagnosed by physician or fasting glucose of ≥126 mg/dL.
NAFLD, non-alcoholic fatty liver disease; OR, odds ratio; CI, confidence interval; TBM%, total body muscle percentage; BMI, body mass index.
a) OR, 95% CI, and p-values were calculated using generalized linear mixed model with logit link function. Each participant was regarded as random effect, and other suggested variables were regarded as fixed effect.
- 1. Kang SH, Lee HW, Yoo JJ, Cho Y, Kim SU, Lee TH, et al. KASL clinical practice guidelines: management of nonalcoholic fatty liver disease. Clin Mol Hepatol 2021;27:363–401.ArticlePubMedPMCPDF
- 2. Bril F, Sninsky JJ, Baca AM, Superko HR, Portillo Sanchez P, Biernacki D, et al. Hepatic steatosis and insulin resistance, but not steatohepatitis, promote atherogenic dyslipidemia in NAFLD. J Clin Endocrinol Metab 2016;101:644–52.ArticlePubMed
- 3. Musso G, Gambino R, Cassader M, Pagano G. Meta-analysis: natural history of non-alcoholic fatty liver disease (NAFLD) and diagnostic accuracy of non-invasive tests for liver disease severity. Ann Med 2011;43:617–49.ArticlePubMed
- 4. Lonardo A, Nascimbeni F, Mantovani A, Targher G. Hypertension, diabetes, atherosclerosis and NASH: cause or consequence? J Hepatol 2018;68:335–52.ArticlePubMed
- 5. Kistner TM, Pedersen BK, Lieberman DE. Interleukin 6 as an energy allocator in muscle tissue. Nat Metab 2022;4:170–9.ArticlePubMedPDF
- 6. Pratesi A, Tarantini F, Di Bari M. Skeletal muscle: an endocrine organ. Clin Cases Miner Bone Metab 2013;10:11–4.ArticlePubMedPMC
- 7. Nara H, Watanabe R. Anti-inflammatory effect of muscle-derived interleukin-6 and its involvement in lipid metabolism. Int J Mol Sci 2021;22:9889.ArticlePubMedPMC
- 8. Cai C, Song X, Chen Y, Chen X, Yu C. Relationship between relative skeletal muscle mass and nonalcoholic fatty liver disease: a systematic review and meta-analysis. Hepatol Int 2020;14:115–26.ArticlePubMedPDF
- 9. Kim Y, Han BG; KoGES group. Cohort profile: the Korean Genome and Epidemiology Study (KoGES) consortium. Int J Epidemiol 2017;46:e20.ArticlePubMedPMC
- 10. Bugianesi E, Bellentani S, Bedogni G, Tiribelli C; Fatty Liver Italian Network. Clinical update on non-alcoholic fatty liver disease and steatohepatitis. Ann Hepatol 2008;7:157–60.ArticlePubMed
- 11. Lee YH, Bang H, Park YM, Bae JC, Lee BW, Kang ES, et al. Non-laboratory-based self-assessment screening score for non-alcoholic fatty liver disease: development, validation and comparison with other scores. PLoS One 2014;9:e107584.ArticlePubMedPMC
- 12. Tennant PW, Murray EJ, Arnold KF, Berrie L, Fox MP, Gadd SC, et al. Use of directed acyclic graphs (DAGs) to identify confounders in applied health research: review and recommendations. Int J Epidemiol 2021;50:620–32.ArticlePubMedPMCPDF
- 13. Kim G, Kim JH. Impact of skeletal muscle mass on metabolic health. Endocrinol Metab (Seoul) 2020;35:1–6.ArticlePubMedPMCPDF
- 14. Zhang H, Lin S, Gao T, Zhong F, Cai J, Sun Y, et al. Association between sarcopenia and metabolic syndrome in middle-aged and older non-obese adults: a systematic review and meta-analysis. Nutrients 2018;10:364.ArticlePubMedPMC
- 15. Zuo X, Li X, Tang K, Zhao R, Wu M, Wang Y, et al. Sarcopenia and cardiovascular diseases: a systematic review and meta-analysis. J Cachexia Sarcopenia Muscle 2023;14:1183–98.ArticlePubMedPMC
- 16. Chung SM, Moon JS, Chang MC. Prevalence of sarcopenia and its association with diabetes: a meta-analysis of community-dwelling Asian population. Front Med (Lausanne) 2021;8:681232.ArticlePubMedPMC
- 17. Beaudart C, Zaaria M, Pasleau F, Reginster JY, Bruyère O. Health outcomes of sarcopenia: a systematic review and meta-analysis. PLoS One 2017;12:e0169548.ArticlePubMedPMC
- 18. Bhanji RA, Narayanan P, Allen AM, Malhi H, Watt KD. Sarcopenia in hiding: the risk and consequence of underestimating muscle dysfunction in nonalcoholic steatohepatitis. Hepatology 2017;66:2055–65.ArticlePubMedPDF
- 19. Wenz T, Rossi SG, Rotundo RL, Spiegelman BM, Moraes CT. Increased muscle PGC-1alpha expression protects from sarcopenia and metabolic disease during aging. Proc Natl Acad Sci U S A 2009;106:20405–10.ArticlePubMedPMC
- 20. Zhang HJ, Zhang XF, Ma ZM, Pan LL, Chen Z, Han HW, et al. Irisin is inversely associated with intrahepatic triglyceride contents in obese adults. J Hepatol 2013;59:557–62.ArticlePubMed
- 21. Miller AM, Wang H, Bertola A, Park O, Horiguchi N, Ki SH, et al. Inflammation-associated interleukin-6/signal transducer and activator of transcription 3 activation ameliorates alcoholic and nonalcoholic fatty liver diseases in interleukin-10-deficient mice. Hepatology 2011;54:846–56.ArticlePubMedPMC
- 22. Zhang J, Xu Y, Lu X, Zhao H. Letter to the editor: NAFLD and sarcopenia: association or causation? Hepatology 2022;76:E119–20.ArticlePubMedPDF
- 23. Chen L, Fan Z, Lv G. Associations of muscle mass and grip strength with severe NAFLD: a prospective study of 333,295 UK Biobank participants. J Hepatol 2022;77:1453–4.ArticlePubMed
- 24. Zhai Y, Xiao Q. The common mechanisms of sarcopenia and NAFLD. Biomed Res Int 2017;2017:6297651.ArticlePubMedPMCPDF
- 25. Kim G, Lee SE, Lee YB, Jun JE, Ahn J, Bae JC, et al. Relationship between relative skeletal muscle mass and nonalcoholic fatty liver disease: a 7-year longitudinal study. Hepatology 2018;68:1755–68.ArticlePubMedPDF
- 26. Lee MJ, Kim EH, Bae SJ, Kim GA, Park SW, Choe J, et al. Age-related decrease in skeletal muscle mass is an independent risk factor for incident nonalcoholic fatty liver disease: a 10-year retrospective cohort study. Gut Liver 2019;13:67–76.ArticlePubMedPMC
- 27. Non-alcoholic Fatty Liver Disease Study Group; Lonardo A, Bellentani S, Argo CK, Ballestri S, Byrne CD, et al. Epidemiological modifiers of non-alcoholic fatty liver disease: focus on high-risk groups. Dig Liver Dis 2015;47:997–1006.ArticlePubMed
- 28. Guo Z, Li M, Han B, Qi X. Association of non-alcoholic fatty liver disease with thyroid function: a systematic review and meta-analysis. Dig Liver Dis 2018;50:1153–62.ArticlePubMed
- 29. Mantovani A, Dalbeni A. NAFLD, MAFLD and DAFLD. Dig Liver Dis 2020;52:1519–20.ArticlePubMed
- 30. Weng G, Dunn W. Effect of alcohol consumption on nonalcoholic fatty liver disease. Transl Gastroenterol Hepatol 2019;4:70.ArticlePubMedPMC
- 31. Antonio J, Kenyon M, Ellerbroek A, Carson C, Burgess V, Tyler-Palmer D, et al. Comparison of dual-energy x-ray absorptiometry (DXA) versus a multi-frequency bioelectrical impedance (InBody 770) device for body composition assessment after a 4-week hypoenergetic diet. J Funct Morphol Kinesiol 2019;4:23.ArticlePubMedPMC
References
Figure & Data
References
Citations


 E-Submission
E-Submission Yeungnam University College of Medicine
Yeungnam University College of Medicine PubReader
PubReader ePub Link
ePub Link Cite
Cite